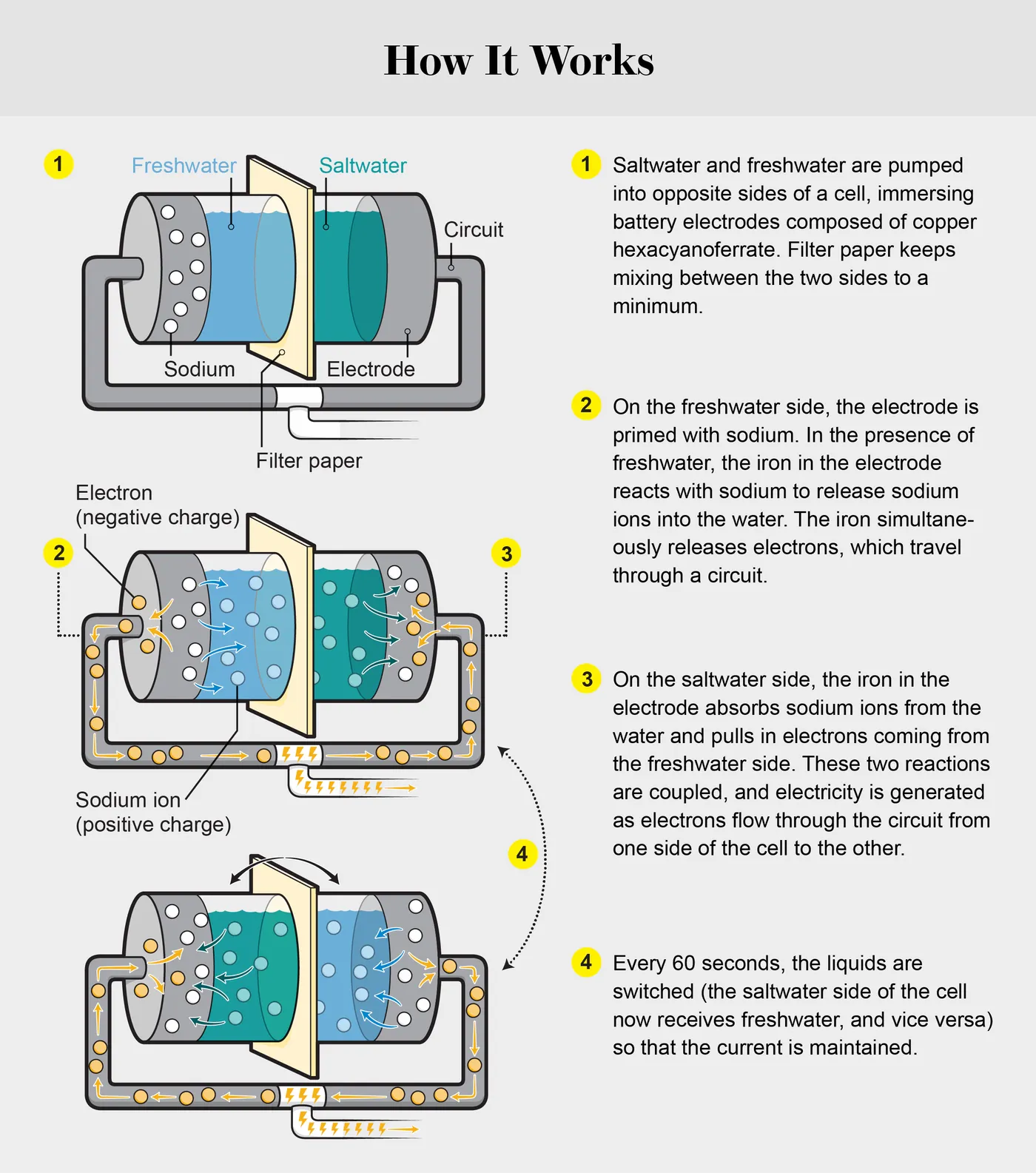
Generating Power from Estuaries
Generating energy from estuaries, the meeting of rivers and oceans, is possible because of the difference in salt concentrations in the water. In fact, scientists estimate that this energy potential is so strong that we could fulfill up to 40% of the world’s electricity needs using this mechanism. However up until now, the existing methods, pressure retarded osmosis (PRO), reverse electrodialysis (RED), and capacitive mixing (CapMix), have been lacking. But never fear, researchers from Penn State have developed a method which combines the strategies of RED and CapMix in an electrochemical flow cell in order to produce greater amounts of energy more efficiently.
Above you can see how the flow cell works. Two channels are separated by an anion-exchange membrane, with a copper hexacyanoferrate electrode in each channel. Graphite foil was to collect the current. From there, one channel is given synthetic seawater and the other channel is given synthetic freshwater. The cell is able to recharge when the water’s flow switches.
"There are two things going on here that make it work," said Christopher Gorski, assistant professor in environmental engineering at Penn State. "The first is you have the salt going to the electrodes. The second is you have the chloride transferring across the membrane. Since both of these processes generate a voltage, you end up developing a combined voltage at the electrodes and across the membrane."
The team determined that stacking multiple cells was able to boost electricity production. As reported in Environmental Science & Technology, with stacked cells, the system produced 12.6 watts per square meter. Compared to previously reported outputs for RED (2.9 watts per square meter) and PRO (9.2 watts per square meter), this combined method is able to generate more power but without some of the problems that the other strategies struggle with.
"What we've shown is that we can bring that power density up to what people have reported for pressure retarded osmosis and to a value much higher than what has been reported if you use these two processes alone," Gorski said.
Nevertheless, a lot more investigation is needed before deciding if this successful prototype could work outside of the lab. Determining if the method is cost-effective and scalable and how it would affect estuary ecosystems is critical before moving on with this technology.