Using Light to Control Organ Development
Optogenetics combined genetic engineering with optics to create a way to control cellular behaviors with light. The pioneers of the technique received a Nobel Prize for their work, which has been continuously refined and advanced. Now investigators at the European Molecular Biology Laboratory (EMBL) have developed a way to use optogenetics to change the shape of developing organs in a fruit fly model. This research, which gives scientists unprecedented control over embryonic development, has been reported in The EMBO Journal.
Cells have to be able to change shape so they can generate the structures required for life. One example of that is in organ development, in which cells are molded in very specific ways and orientations. Project leader Stefano De Renzis and his colleagues are investigating the mechanisms underlying shape changes in cells, and are using optogenetics in to manipulate those changes.
In the video below (courtesy of Daniel Krüger / EMBL), from the side, you can see the outer surfaces of cells contracting, while the inner side relaxes. That coordinated movement creates a force that pushes the cell to the interior of the embryo, an initial step in organ development called invagination. Internal organs have to be just that - inside, so clumps of cells must move there to properly form the animal. During invagination, a surface formed by a group of cells folds inwards as it contracts.
"Imagine the embryo as a balloon and tissue invagination as the deformation caused by fingers that push the surface of the balloon inwards. The only difference is that cells are not being subjected to an external force like the fingers, but need to be able to generate forces to move inside by themselves," said De Renzis. When these processes go awry, it can wreak havoc in the organism.
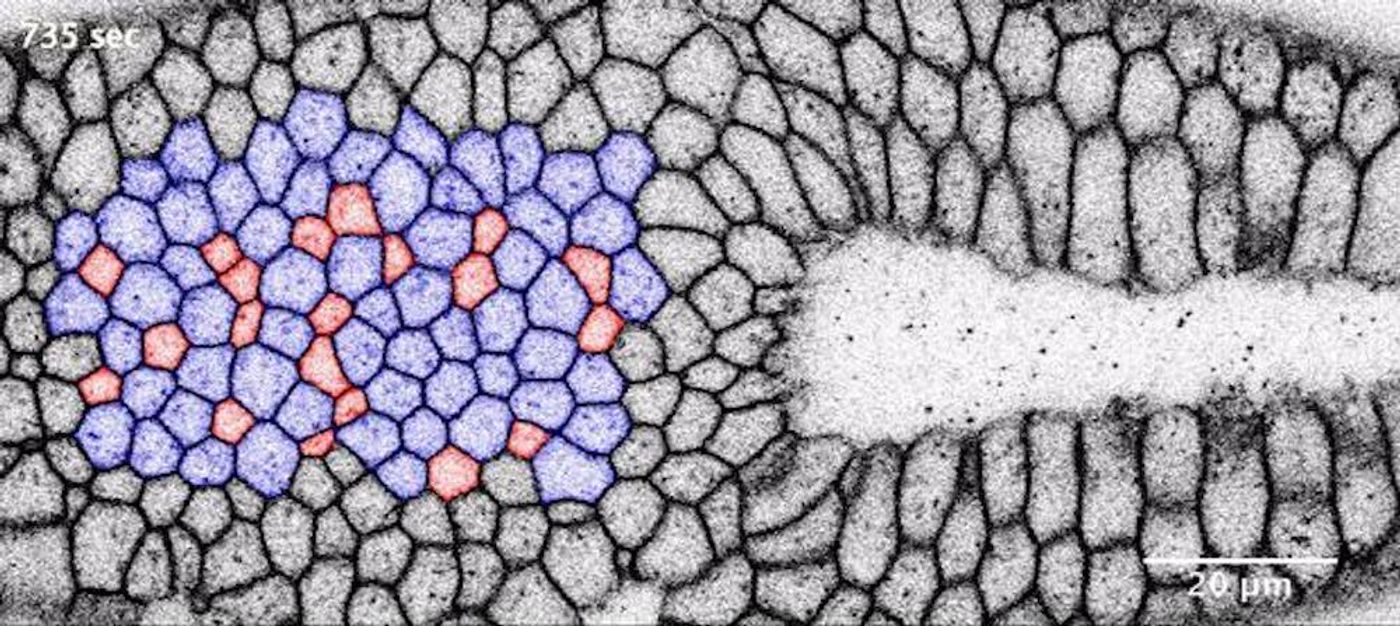
By manipulating the natural processes of invagination, the research team can learn more about how it works. One aspect to alter is the malleability of those moving cells. When optogenetic tools are used to stiffen the cell surfaces, they cannot bend inwards, and invagination halts.
"If cells are not allowed to relax their bases, they cannot constrict their apices efficiently, and tissue invagination stops. To stick with the balloon analogy, it's like when you squeeze the top and the bottom of a balloon simultaneously. The inner pressure becomes higher, and the balloon can't fold inwards anymore," explained De Renzis. Optogenetics has allowed the scientists to stop invagination before or while it happens.
In the video below (courtesy of Daniel Krüger / EMBL), modified and natural invagination can be visualized. Optogenetically modified tissue on the left won’t invaginate, while on the right, natural invagination forms a pouch.
The researchers were able to learn more about the inner (basal) surface of tissues with these new techniques, which didn’t do damage to the cells. This work provides insight into how abnormalities might arise during embryonic development.
Though the findings are from fruit flies, De Renzis thinks that these basic biological processes work in very similar ways in other organisms. Optogenetics has provided scientists with the ability to answer some long-standing questions. It may also help them create artificial tissues, or to harness the process for regenerative medicine.
Sources: AAAS/Eurekalert! via EMBL, EMBO Journal


![WGS for rare disease diagnosis [eBook]](https://d3bkbkx82g74b8.cloudfront.net/eyJidWNrZXQiOiJsYWJyb290cy1pbWFnZXMiLCJrZXkiOiJjb250ZW50X2FydGljbGVfcHJvZmlsZV9pbWFnZV84MmRlM2UyYjA5M2Q3ZTYwOTI3Zjc1YTRjOWU2N2RmMjkzMThjMTJkXzI1MDcucG5nIiwiZWRpdHMiOnsidG9Gb3JtYXQiOiJqcGciLCJyZXNpemUiOnsid2lkdGgiOjcwMCwiaGVpZ2h0IjozNTAsImZpdCI6ImNvdmVyIiwicG9zaXRpb24iOiJjZW50ZXIiLCJiYWNrZ3JvdW5kIjoiI2ZmZiJ9LCJmbGF0dGVuIjp7ImJhY2tncm91bmQiOiIjZmZmIn19fQ==)





