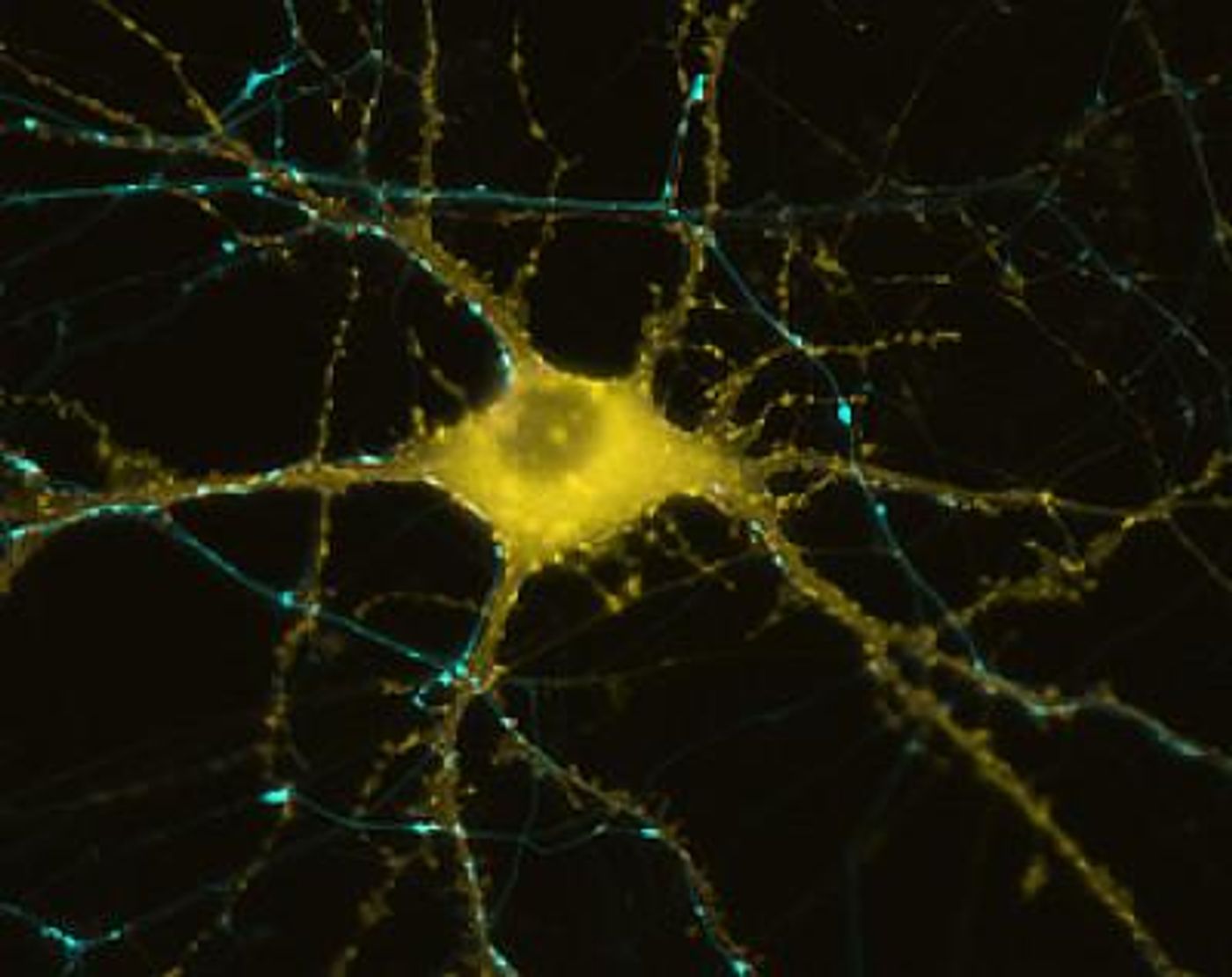
Modified CRISPR Can Manipulate Gene Activity in Neurons
There are challenges to working with nerve cells in the lab, which can create research bottlenecks in the study of neurological disease. Human nerve tissue is difficult to acquire, and even then, neurons can’t usually be grown in the lab for long periods of time like other types of cells. Nerve cells can, however, now be created using a recently developed, Nobel-prize winning technology that genetically reprograms cells to be stem cells. These induced pluripotent stem cells can then be directed to create any cell type, including neurons. Applying the CRISPR/Cas9 gene editing technology was thought to usher in a new era for these cells, but first, more problems had to be overcome.
"Prior to this study, there were significant limitations that restricted what scientists could do with human neurons in the lab," said Martin Kampmann, PhD, associate professor in UCSF's Institute for Neurodegenerative Diseases and co-senior author of a new study in Neuron (open access at bioRxiv) that merges the CRISPR tool with neurons made from induced pluripotent stem cells.
"It was possible to get neurons donated by patients who had undergone procedures that involve removing brain tissue to treat epilepsy or brain cancer. But these samples can only survive for a few days. You can't perform experiments to probe gene function on short-lived neurons," Kampmann added.
The CRISPR system relies on an enzyme that cuts DNA called Cas9, and stem cells were found to be quick at repairing that damage.
"Stem cells have a very active DNA damage response. When Cas9 produces even just one or two DNA cuts, it can lead to toxicity that causes the cells to die," Kampmann explained.
As a postdoctoral fellow in UCSF Professor Jonathan Weissman’s lab, Kampmann helped create a tool called CRISPRi (described in the video). CRISPRi can interfere with the activity of a gene without cutting DNA, so it will work in neurons derived from stem cells, and is not toxic.
The scientists tested their system to learn more about the genes that influence the development of brain disease. Some genes can extend the life of neurons, for example, or increase the number of projections and branches that neurons produce.
A surprising finding regarded so-called housekeeping genes, which are thought to be essential genes that behave in basically the same way in most cells. The scientists disrupted the same housekeeping gene in neurons and stem cells, and observed totally different impacts. The two different types of cells changed the genes they activated in different ways, which suggests that these housekeeping genes may actually be doing very different things in various cells. The researchers want to investigate further.
They are also using their tools to study diseases that selectively affect certain kinds of neurons. Kampmann wants to know more about how this technique will work in other kinds of brain cells. The ultimate goal is to create therapeutic interventions for brain disorders.
"One of the big challenges facing the field is that, for most of these disorders, the precise molecular pathways that we should target for drug development remain unclear," said co-senior study author Michael Ward, MD, Ph.D., a physician-scientist at the National Institutes of Health.
"With this technology, we can take skin or blood cells from a patient with a neurodegenerative disease like Alzheimer's, turn them into neurons or other brain cells, and figure out which genes control the cellular defects associated with the disease," said Kampmann. "The information may allow us to identify effective therapeutic targets."
Sources: AAAS/Eurekalert! Via UCSF, Neuron