Brown Fat Transplants Could Reduce Metabolic Disease
The Centers for Disease Control and Prevention (CDC) estimates that in 2018, obesity impacted about 42 percent of American adults. People with obesity are at higher risk for many diseases, including type 2 diabetes, coronary heart disease, and stroke; they also tend to generally have a lower quality of life and a higher risk of death. As of 2008, obesity was thought to cost the United States about $147 billion a year. All this makes obesity an intense focus of research studies.
Scientists are now learning more about how the different types of fat in the human body are involved in metabolic disease. It's known that brown fat cells work to burn energy, instead of storing it as white fat cells do. If there was a way to add brown fat or convert white fat into brown fat it could help reduce the prevalence of obesity.
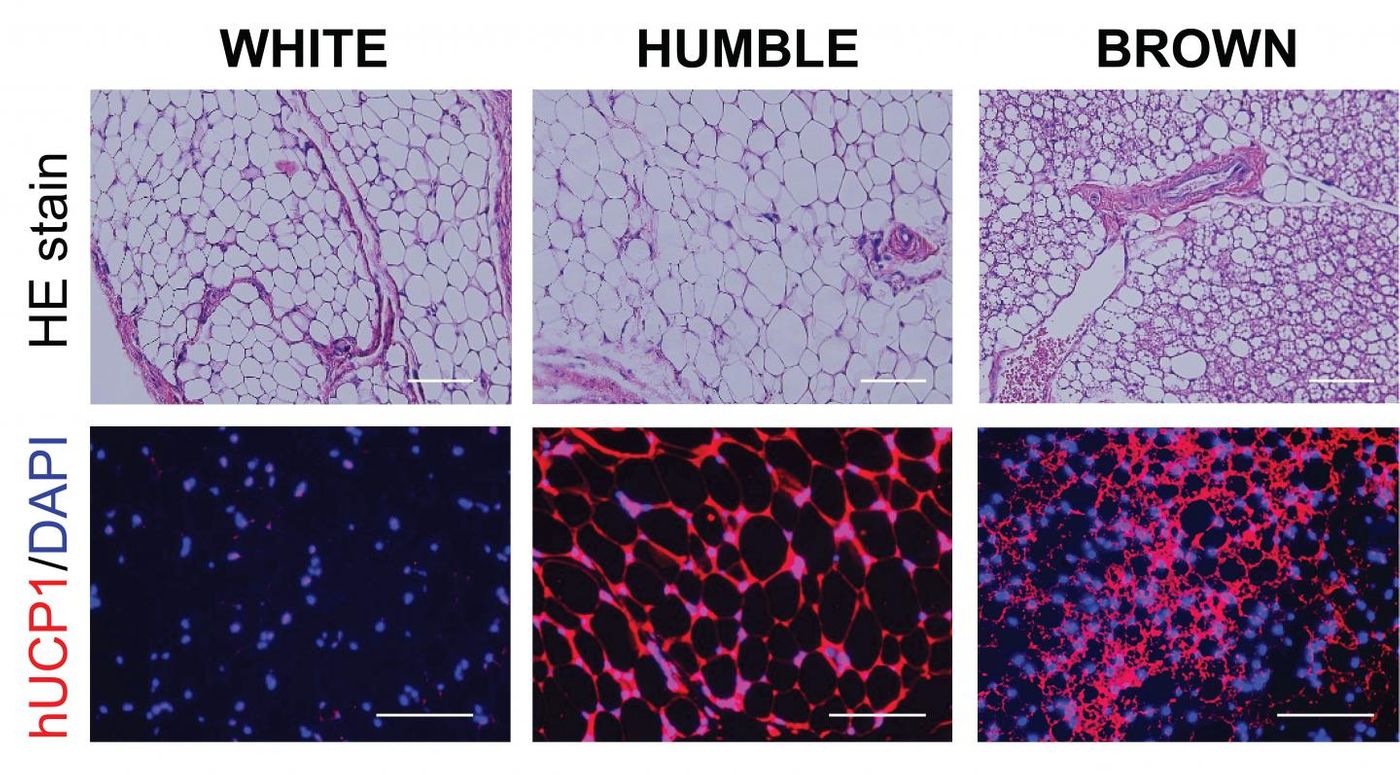
Researchers have now developed a way to genetically modify human white fat cells with CRISPR technology so they are more similar to brown fat cells. These HUMBLE (human brown-like) fat cells that generate heat instead of storing energy could then be transplanted into patients with obesity as a therapeutic, said Yu-Hua Tseng, Ph.D., a senior investigator in Joslin Diabetes Center's Section on Integrative Physiology and Metabolism. Tseng is the senior author of the report on this work in Science Translational Medicine.
These newly-transplanted brown fat cells would then theoretically be able to reduce the levels of excess glucose and lipids in the blood, which is a characteristic of metabolic diseases like diabetes. People that are overweight or obese often have less beneficial brown fat, and the HUMBLE cells can help them overcome this problem, Tseng said.
The HUMBLE cells were made by using human white fat cells that are not fully differentiated; they are in a progenitor state and not in their final form. With CRISPR-Cas9, the researchers raised the expression of a gene called UCP1 to cause the progenitor white fat cells to become like brown fat cells.
The scientists transplanted these cells into a mouse model that lacked an immune system, reducing the likelihood of rejection, and the cells seemed to function like brown fat cells. Mice with the transplanted cells had greater sensitivity to insulin and were better at removing glucose from their blood, and they gained less weight than mice that had received transplanted white cells.
The scientists found that any health benefits were primarily due to transplanted cells to signaling to existing brown fat cells in the mice. "Cells in different tissues communicate with each other," Tseng said. "In this case, we found that our transplanted HUMBLE cells secrete a molecule called nitric oxide, which is carried by red blood cells to the endogenous brown cells and activates those cells."
This therapy is not ready for the clinic, but Tseng is hopeful that if it works in pre-clinical studies, it may one day be possible to apply this technique to individual patients. In that process, a small number of cells would be removed from a patient, isolated and modified, then returned to the patient as HUMBLE cells. Tseng's team is also interested in a more generally applicable and less costly approach. For example, it may be possible to use generic cells that are not removed from the patient, by coating them in a material that would protect them from the immune system.
"Employing cell-based or gene therapies to treat obesity or type 2 diabetes used to be science fiction," she says. "Now scientific advances, such as CRISPR gene-editing technologies, will help us to improve the metabolism, the body weight, the quality of life, and the overall health of people with obesity and diabetes."
Sources: AAAS/Eurekalert! via Joslin Diabetes Center, Science Translational Medicine