Using CRISPR to Destroy Cancer Cells
A few days ago, researchers and Professors Jennifer Doudna and Emmanuelle Charpentier won the Nobel prize in Chemistry for their work developing the gene editor CRISPR/Cas9. Since it was created, the gene editor has been used in many ways in research labs and has even moved to human trials in the clinic.
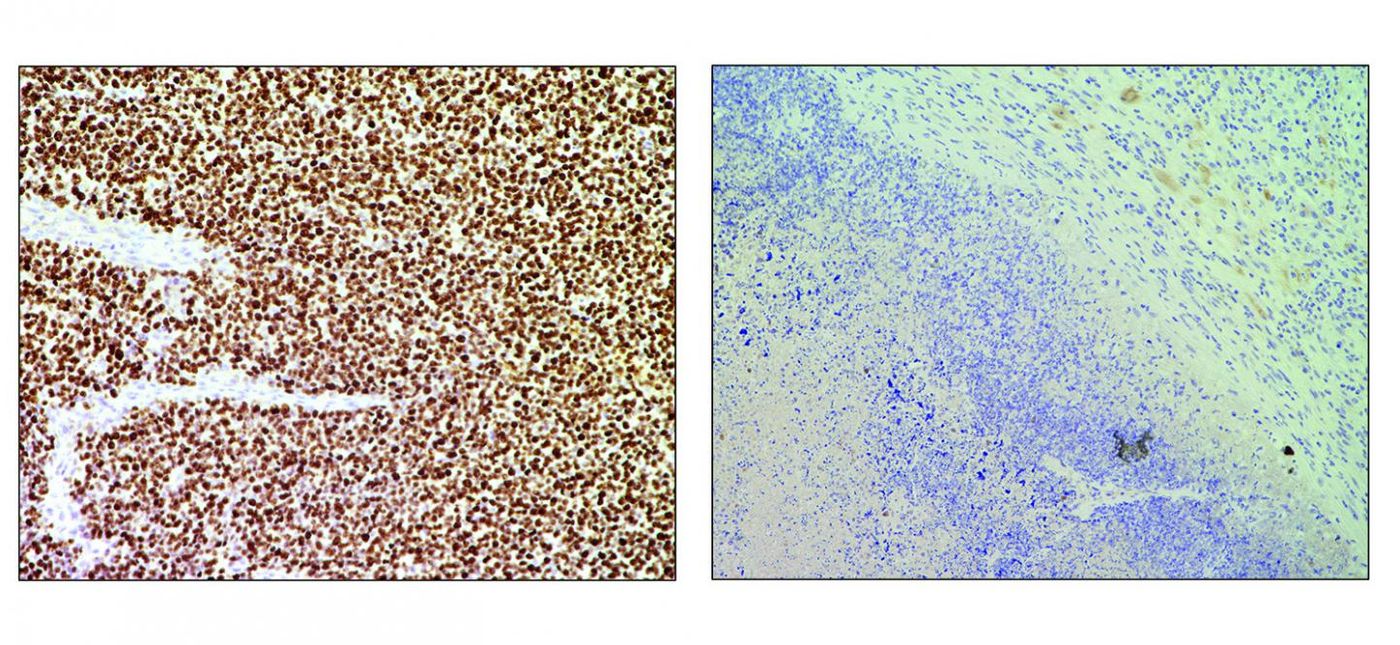
Some diseases can be traced back to genetic errors, and in some cases, those errors have the potential to be repaired by the CRISPR/Cas9 system. In new research, scientists have now applied it to tumor cells in an effort to eliminate something called fusion genes; fusion oncogenes are only found in cancer cells. The work has been reported in Nature Communications.
Fusion genes come from the aberrant joining of fragments of DNA that arise from two different genes, an error that can occur during cell division. Cells that carry deleterious fusion genes will often simply die, but in some cases, the cell is unharmed by the fusion gene; the cell carrying the fusion gene may have a reproductive advantage that causes it to start multiplying and leads to the formation of a tumor in the case of fusion oncogenes.
"Many chromosomal rearrangements and the fusion genes they produce are at the origin of childhood sarcomas and leukemias," noted the lead co-author of the study, Sandra Rodríguez-Perales.
Fusion genes have been found in many tumor cells; they may be present in up to twenty percent of cancers including breast, brain, and lung tumors. Fusion oncogenes are an attractive therapeutic target because they’re not found in healthy cells.
CRISPR/Cas9 can target very specific places in the genome for edits. In this work, the researchers used it to remove fusion genes from cells and mouse models of both Ewing's sarcoma and chronic myeloid leukemia. The researchers tried to make the tumors destroy themselves.
"Our strategy was to make two cuts in introns, non-coding regions of a gene, located at both ends of the fusion gene," explained study co-author Raúl Torres-Ruiz. "In that way, in trying to repair those breaks on its own, the cell will join the cut ends which will result in the complete elimination of the fusion gene located in the middle.” This particular gene is required for the cell to survive, so once the repair was made, the tumor cell died.
"Our next steps will be to carry out more studies to analyze the safety and efficiency of our approach," added Rodríguez-Perales. "These steps are essential to know if our approach can be translated in the future into a potential clinical treatment. Furthermore, we will study whether our strategy, which we have already seen works in Ewing's sarcoma and chronic myeloid leukemia, is also effective in other types of cancer caused by fusion genes and for which there are currently no effective therapies."
Sources: AAAS/Eurekalert! via Centro Nacional de Investigaciones Oncológicas (CNIO), Nature Communications