Super-Spreader Events Promoted 2018-2019 Hantavirus Outbreak
We've all learned about super-spreader events over the past year, but occasions where a large group of people gathers and many people become infected with a pathogen are not restricted to the COVID-19 pandemic. Scientists have investigated an outbreak of hantavirus pulmonary syndrome (HPS) that happened in 2018 and 2019 in a small Argentinian village and found that extensive contact between people and super-spreader events encouraged it. The findings have been reported in the New England Journal of Medicine.
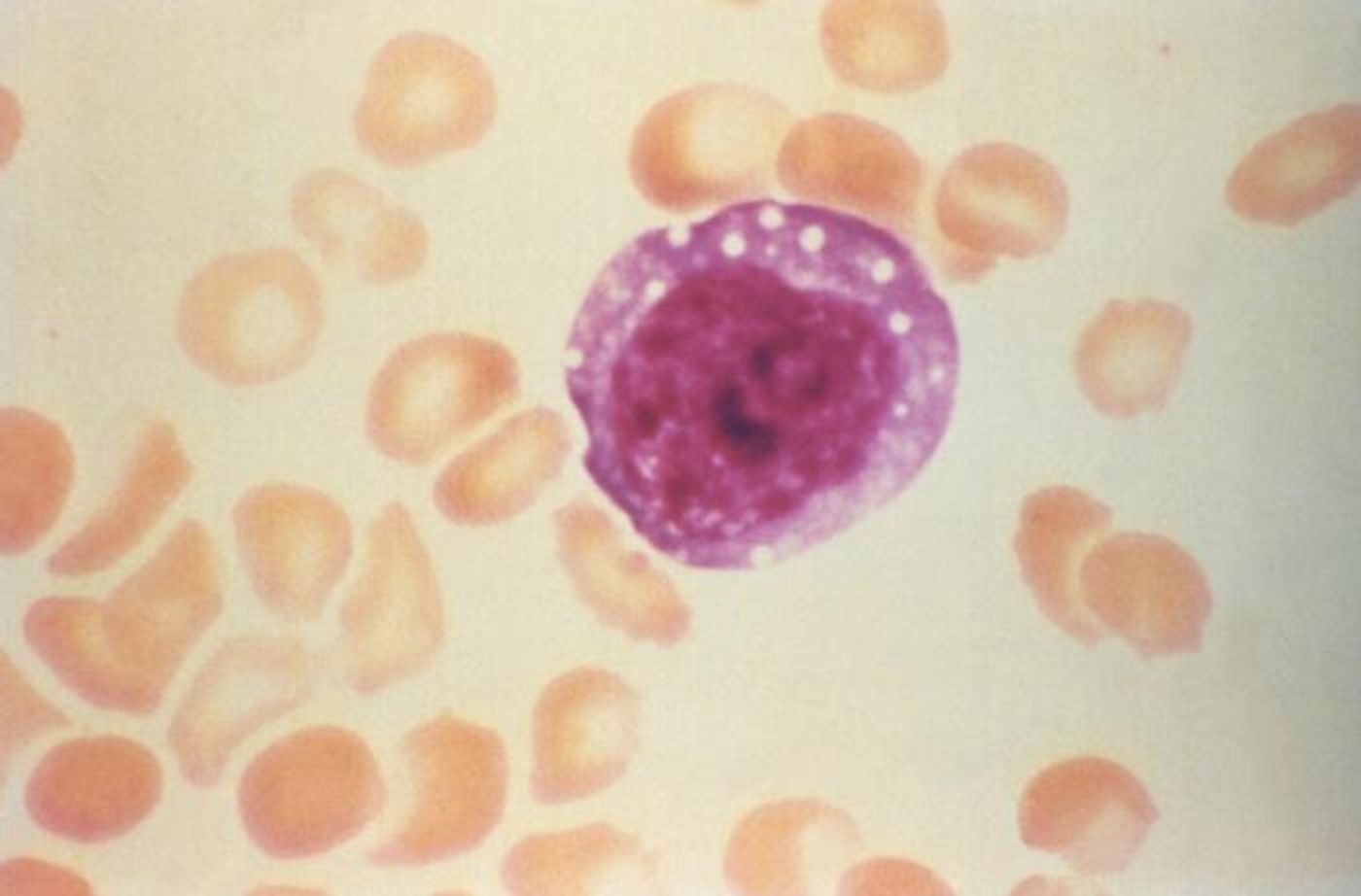
Rodents carry hantaviruses, and in South America, they carry a hantavirus called Andes virus, which can be transmitted to people if they are exposed to infected animals or their droppings. Andes virus can also be transmitted from one person to another, the only known hantavirus to do so. It may spread through direct contact with an infected person or their bodily fluids, or simply by being near a person sick with the virus. Infected individuals are at risk for a serious respiratory condition called Hantavirus Pulmonary Syndrome (HPS), which can be fatal. There are no treatments or vaccines at this time.
In this study, researchers analyzed the genetic, clinical, and epidemiological characteristics of the outbreak. It may help prevent or manage similar events in the future.
The study determined that large social events and people with high viral loads propelled the transmission of Andes virus, which caused 34 confirmed cases. The virus had a 32 percent fatality rate. This is the most extensive person-to-person transmission that's been documented for this disease. Aerosolized particles might have been a route of transmission, noted the researchers. It took anywhere from nine to forty days for symptoms to appear, though they also added that the window of infection may be more limited. Transmission seemed to happen on the first day of a patient's fever. The researchers did not find an explanation for why the virus was severe in some but not others.
"This important observation indicates that, regardless of the severity of the disease, all HPS cases should be equally managed concerning their potential for virus transmission," said co-senior study author Gustavo Palacios, Ph.D., Division Chief of Molecular Biology at U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID). "Furthermore, we need to re-evaluate the threat that this virus poses. The lack of existing medical countermeasures, super-spreading potential, and high case-fatality rate all should raise concern."
Genomic 'signatures' indicated that the virus that caused the 2018-2019 outbreak was strikingly similar to one that caused an outbreak in 1996. This genetic data may help researchers create a vaccine or treatment.
The study also showed evidence of dysfunctional immune regulation in patients. "We hope that by further exploring this apparent immune dysregulation, we will expand our knowledge of how different hantavirus species cause hantavirus pulmonary syndrome," said study co-author Mariano Sanchez-Lockhart, Ph.D., of USAMRIID. "Gaining a better understanding of how our immune system tackles Andes virus infection will certainly help clinicians to better manage severe cases and pave the way for new therapeutic interventions."
"We hope this approach serves as a model for future investigations of person-to-person transmission and for the identification of super-spreading events," said study co-author Peter Larson, Ph.D., of USAMRIID.
Sources: AAAS/Eurekalert! via US Army Medical Research Institute of Infectious Diseases, New England Journal of Medicine