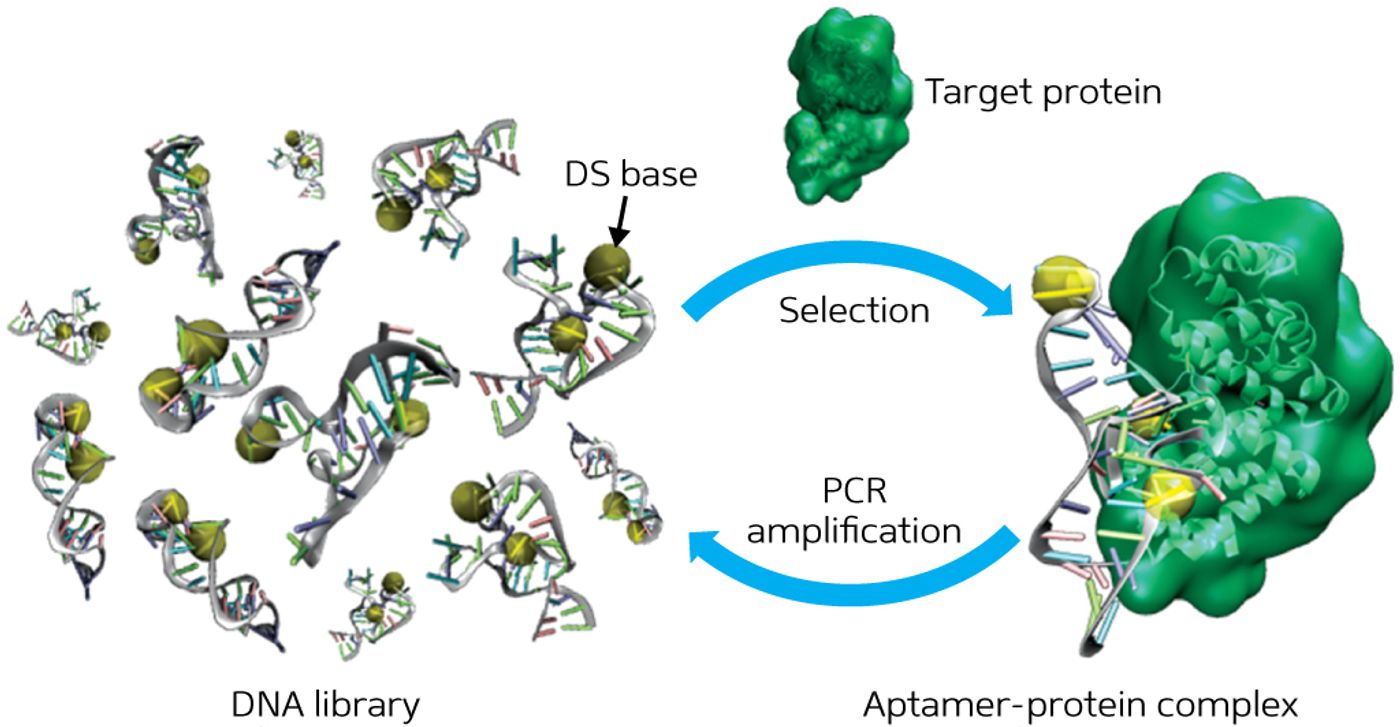
Single-stranded molecules of DNA called “aptamers” have been studied since 1990 for their ability to bind and inhibit molecular targets, a useful behavior for disease detection and drug delivery (
Nature). However, aptamers have not been approved for clinical use yet because of variable binding ability and quick digestion by enzymes. With two modifications, though, a team of researchers at the Institute of Bioengineering and Nanotechnology (IBN) at the Agency for Science, Technology and Research (A*STAR), could soon be on the way to developing the first approved aptamer technology.
Just like the antibodies produced by the immune system that target foreign pathogens, aptamers have the potential to become a specialized army for infectious diseases. However, just like medicine, aptamers need time in the bloodstream to find and bind to their target. If they are quickly and easily digested by enzymes waiting in the blood stream, the aptamers will not have time to act, making their effectiveness irrelevant.
IBN Principal Research Scientist and Team Leader Dr. Ichiro Hirao and a team of researchers from A*STAR equipped their aptamers with a sequence of DNA called a “mini hairpin DNA” to prevent enzyme digestion, allowing the aptamers time to find and inhibit their targets. With the mini hairpin DNA, aptamers can survive for days rather than hours, as the DNA sequence acts as a “protective shield.”
Hirao’s team also overcame the problem of aptamer binding capabilities by adding an “unnatural base” that improved binding by 100 percent. Unnatural bases were developed to supplement the natural bases – A, T, G, and C (
Nature). In this case, the extra component of the single-strand of DNA greatly enhanced its binding capabilities.
We hope to use our DNA aptamers as the platform technology for diagnostics and new drug development," said IBN Executive Director Professor Jackie Y. Ying. Scientists from the IBN plan to soon begin testing the aptamers for specific diseases. Their current study is published in the journal
Scientific Reports.
Source:
A*STAR