Structure of Critical DNA Repair Protein Revealed
As we age and whenever tissue is damaged, cells divide and replace old ones. Every time, the genetic material in the nucleus has to be accurately replicated. Every replication event has to be free of errors, a tall order for something that happens so often to a huge molecule like DNA. When those errors do inevitably happen, they must be repaired in order to keep the cell alive and healthy. Scientists have now revealed the structure of a protein that is critical to that process.
ATR kinase steps in at the first sign of damage to DNA, calling up the repair mechanisms of the cell. Now researchers know more about how it acts, reporting their findings in Science.
"The ATR protein is the apical kinase to cope with the DNA damages and replication stress," explained the lead author of the work, Gang Cai, a Professor of Life Sciences at the University of Science & Technology of China in Hefei, China. "It has long been a central question to determine [the] activation mechanism of ATR kinase--how it responds to DNA damage and how it is activated."
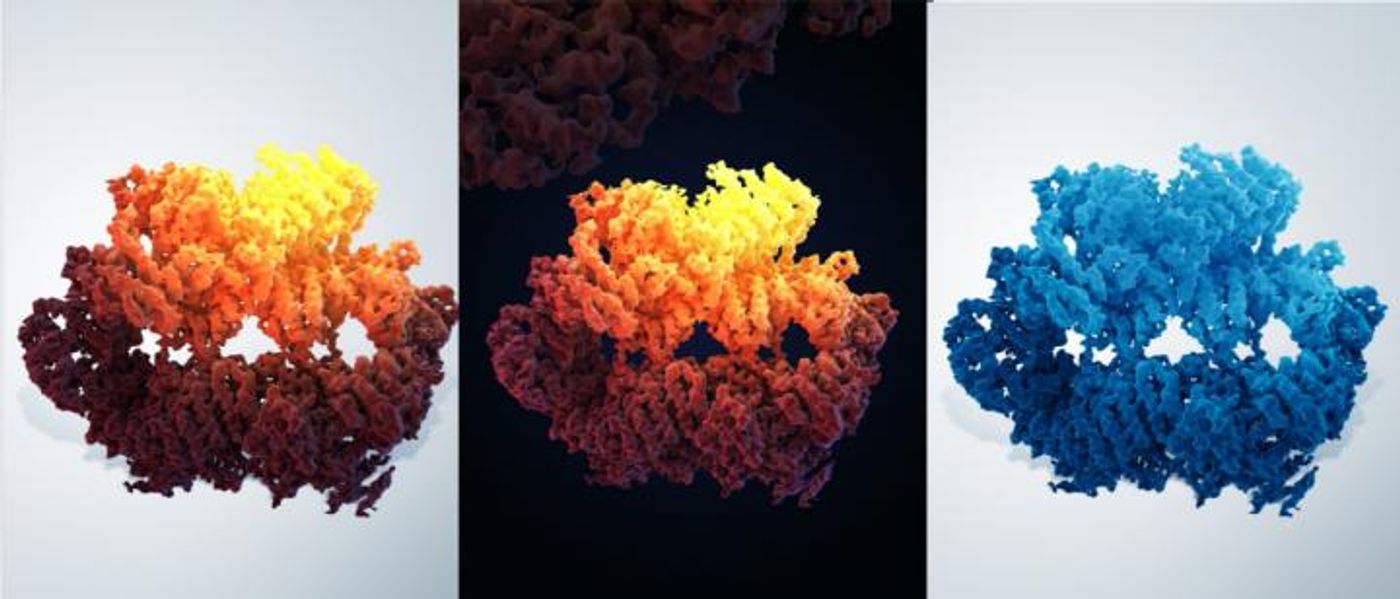
Cai and colleagues utilized electron microscopy to visualize the Mec1-Ddc2 complex at 3.9 ångströms, which is roughly eight times bigger than a single atom of helium. That complex can be found in yeast, and it is considered to be equivalent to the human ATR protein and ATRIP, its partner in cell signaling.
As one of six proteins that maintain the health of the cell, the ATR kinase is critical. When it or another member of its family of proteins identifies a problem like DNA damage, they initiate the chemical cascades that are necessary to fix the problem.
"Cryo-electron microscopy of the Mec1-Ddc2 with state-of-the-art instrumentation has resulted in an electron density map at near-atomic resolution," noted Cai, who added that the improved map has confirmed added to previous findings.
ATR has been considered a potential target for therapeutics for a long time. The new, high-resolution structural data provided by Cai and his team have identified sites of ATR kinase regulation. Understanding more about how the kinase is regulated will aid scientists who are developing new therapeutics based on this system.
"The structure of [the] yeast member closely resembles those of the human counterpart," said Cai, who noted the significant similarity in the detailed architecture. "We believe the information acquired from the yeast Mec1-Ddc2 [sheds] light on the architecture and mechanism of [the] human ATR-ATRIP complex."
Cai and his team are continuing to build on this work, imaging the yeast Mec1-Ddc2 and its human counterpart at various activation points. They want to develop more efficient ATR inhibitors that are more specific and may help improve cancer treatments.
Hear more about how molecules that target ATR can be used in cancer therapeutics from the video above.
SOurces: AAAS/Eurekalert! Via University of Science and Technology of China, Science