Researchers find that obesity has a paradoxical effect on Cancer
Cancer therapy works differently in different people. Understanding what effects the individual body’s response to treatment will be crucial for the development of patient-centered therapies. Obesity is one of the confounding factors for cancer treatment.
Findings published by UC Davis researchers today in Nature Medicine provides new evidence on understanding the role of obesity in tumor growth and immunotherapy efficacy. It aids in the tumor growth and allows for enhancing the tumor-targeted immunotherapies. Watch, the video below to learn more about how immunotherapies work in cancer treatment.
"It's counter-intuitive because up to this point all of our studies showed that obesity resulted in more toxicities associated with immunotherapy treatments," said William Murphy, a co-last author of the study and vice chair of research in the UC Davis Department of Dermatology. "This is a game-changer because when we personalize medicine and look at body mass index, in some situations, it can be bad, and in some situations, it can be helpful."
A retrospective, multi-cohort study in patients with metastatic melanoma, published in February this year, showed that obesity improved the patient survival with targeted immune therapy. However, the underlying mechanism of action has not to be investigated.
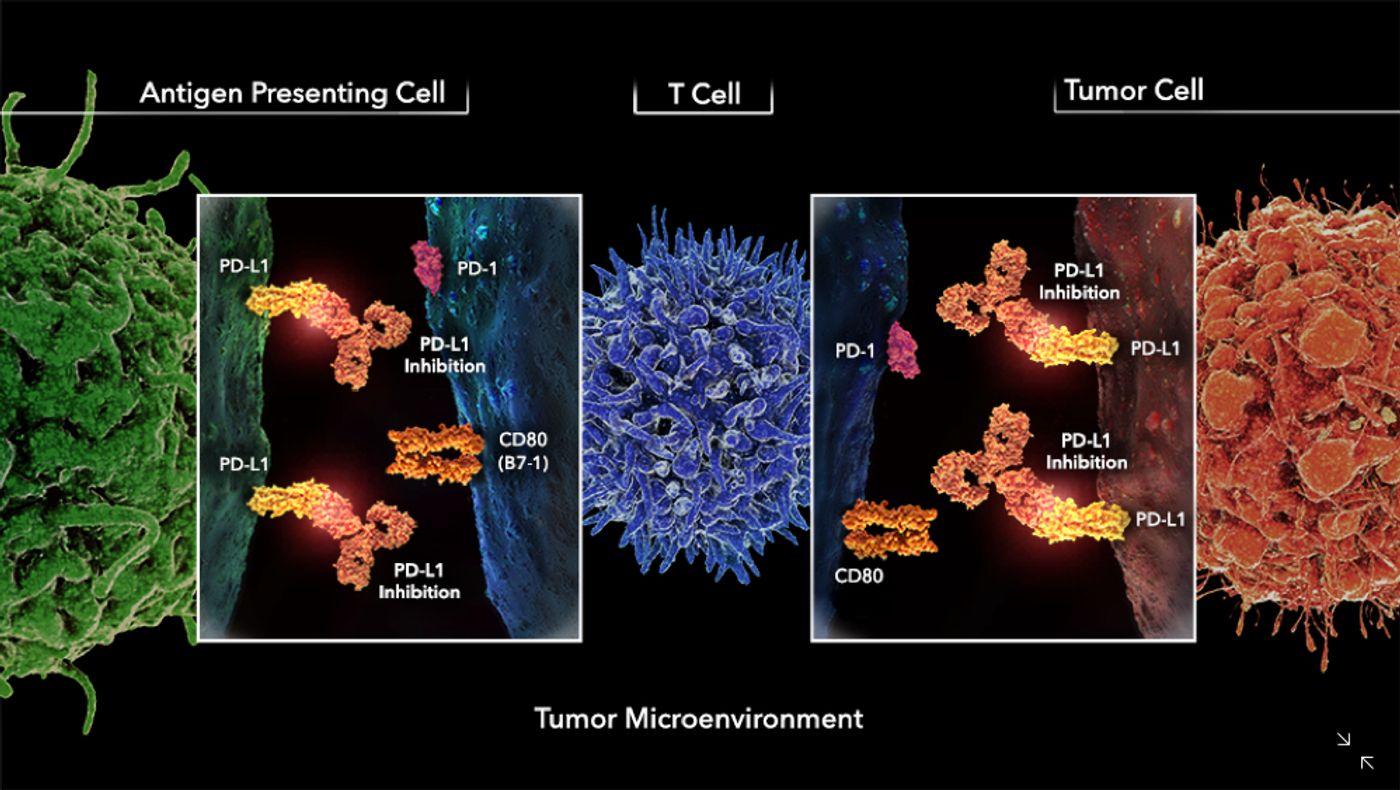
Ziming Wang, the lead author on the manuscript published, investigated the impact of obesity on T cell responses, tumor progression with PD-(L)1 checkpoint blockade immune therapy. These drugs work by blocking pathways called immune checkpoints that cancers use to escape the immune system. They include drugs like Keytruda (pembrolizumab), which have dramatically improved survival in many lung cancer and melanoma patients. The contradictory effects of fat on cancer therapy was confirmed from studies in mice, nonhuman primates and humans.
Image: PD-1: programmed cell death-1, Credit: Azimmuno-Oncology
T cells across all species demonstrated an increase in PD-1 expression in obese subjects when compared to nonobese subjects. Transcriptome analysis of the T cells from tumor-bearing obese mice was markedly different from tumor-bearing nonobese mice and supports increased cell exhaustion and inflammation. Furthermore, obesity promoted tumor growth across species.
"In obese animals, cancer grows faster because there are more nutrients for tumors and because the immune system is more suppressed," said Murphy.
A meta inflammatory state is observed in obese cancer patients, with increased proinflammatory cytokines, glucose, insulin, fatty acids, and leptin. RNA sequencing of the T-cells showed the role of leptin, a weight-regulating hormone produced by the body's fat cells, in particular on fat metabolism and T cell signaling. Healthy human blood analysis showed an increased serum leptin levels in obese donors, and it correlated with a PD-1 expression on T cells. The study provides evidence from in vitro and in vivo studies across multiple species to support the association of leptin with PD-1 expression and T cell exhaustion.
The PD-1 blockade was found to be more effective in tumor regression in obese mice than in control of nonobese mice. These antitumor observations seen in mice models also extended to human patients. A study done on a large cohort of 152 human colorectal cancers, showed a marked decrease in tumor-infiltrating T cells in obese patients. Additionally, studying the PD-1 expression in 251 patients with melanoma when treated with PD-1 blockade therapy mimic the results from mice and striking improvement in the clinical outcomes were seen in obese patients.
"Overall these findings suggest that obesity may be a very important biomarker for response to checkpoint inhibitor immunotherapy," said Arta Monjazeb, associate professor in the UC Davis Department of Radiation Oncology and co-last author on the study. "We are not advocating for obesity as improving the prognosis for cancer patients. But obesity appears to induce immune suppression and accelerated tumor growth through mechanisms that can be successfully reversed by checkpoint inhibitor immunotherapy."
Murphy cautioned that while the findings are an essential step in better targeting immunotherapies, many other factors likely influence how effective a specific drug will be for a given patient. Those may include gender, the type of diet consumed, their microbiome and the timing of their treatment.
"So much data has been accumulated on understanding how factors in the tumor can affect response to immunotherapy," he said. "We are now opening the door to understanding how intrinsic factors such as age, sex, diet, and obesity also can affect the immune system and response to immunotherapy."
The video below by Dr. Arta Monjazeb, a co-last author, discusses the research at UC Davis that led to the surprising findings.
Sources: AAAS and EurekAlert!, Nature Medicine, The Lancet Oncology, UC DAVIS HEALTH