Flu Shot Less Effective Due to Overuse of Antibioitics
New research out of the Stanford University School of Medicine shows that the consequence of overuse of antibiotics lowers the effectiveness of the seasonal flu vaccine. It is well established that antibiotic use takes a significant toll on the diversity and the overall population of the microbiome. Now it seems that depletion may affect how the human body reacts to the flu vaccine.
Back in 2011, Professor of Microbiology and Immunology at the Stanford University School of Medicine, Dr. Bali Pulendran, experimented with mice with an undeveloped microbiome to see how they would react to antibiotics. The lab raised mice in a sterile environment, making their intestinal tract germ-free, then the mice were given antibiotics. Results clearly showed that mice without bacteria in their gut struggled to process antibiotics, while mice with a normal, diverse, microbiome were able to work with the antibiotic.
Like much of the research published about the importance of a diverse microbiome, the 2011 study only proved the concept in mice. The implication is that humans with a weak microbiome would similarly struggle to process antibiotics. In 2014, Dr. Pulendran set out to test this implication in a clinical trial.
Over the 2014-2015 flu season, Dr. Pulendran's lab conducted a clinical trial to verify if the findings in mice applied to humans. Twenty-two adults aged 18-45 years old were split into two equal groups. The first group received "broad-spectrum" antibiotics. The treatment included metronidazole, commonly used to treat rosacea and yeast infections, and vancomycin, which is used to treat staph infections. The group took antibiotics for five days, and on day four received the seasonal influenza vaccine. The second group received no antibiotic treatment and were also given a flu shot on day four of the study.
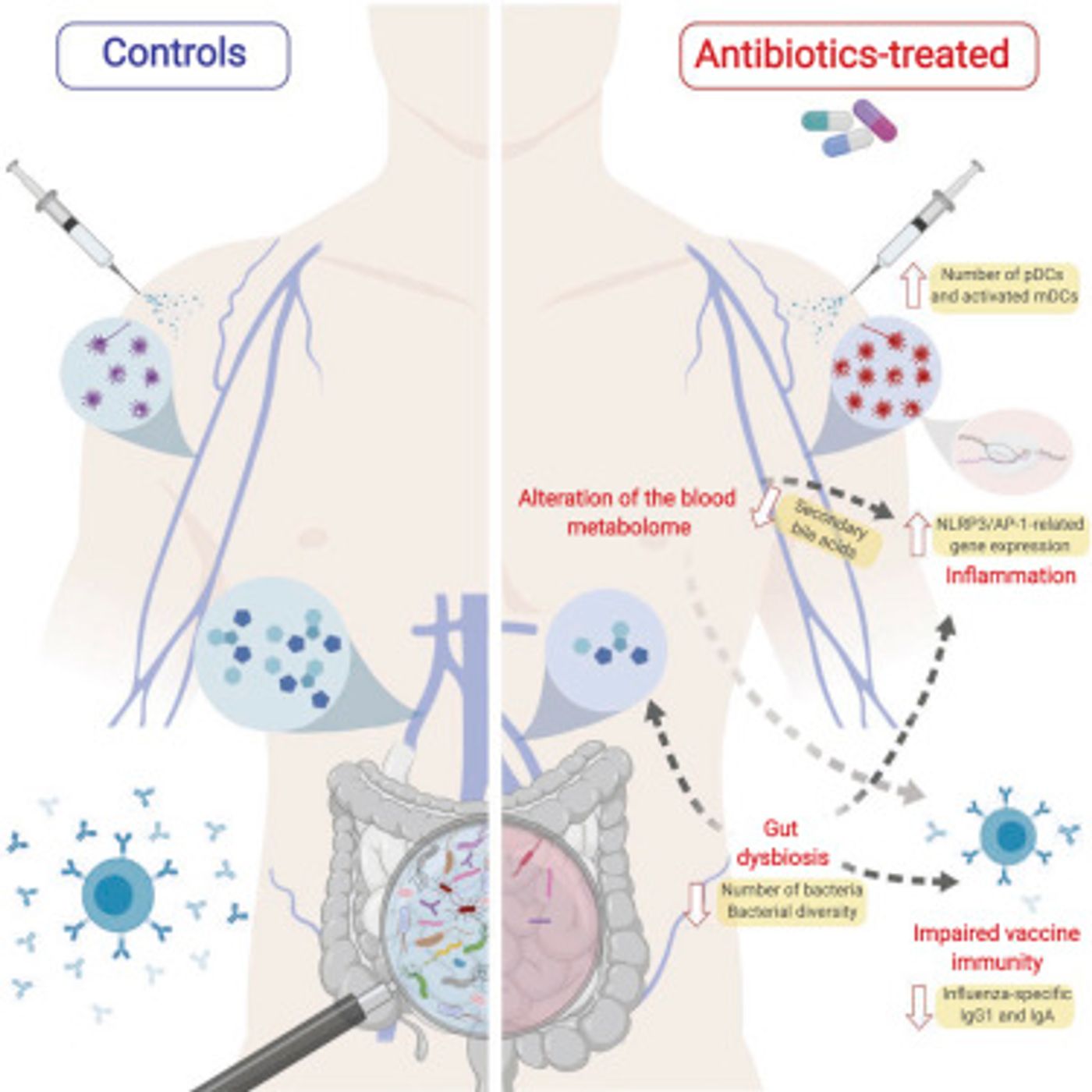
Researchers found that the antibiotics decreased individual's gut-bacteria population by 10,000-fold, or the equivalent of the human population plummeting to 700,000. Interestingly, the effects of antibiotics on the number and diversity of gut-bacteria were measurable for up to a year after antibiotic treatment.
However depleted the microbiome was, all twenty-two individuals showed comparable increases in influenza antibodies 30-days after their flu shot. The baseline number of influenza antibodies in this group of individuals was relatively high because most participants had either received a flu shot or had the flu in recent years.
In the following flu season, the researchers reran the clinical trial with 11 similarly aged volunteers, but all with a low number of influenza antibodies. None volunteers for this trial received a flu shot in the last three years, so their immune system has fewer influenza antibodies. Five individuals were given the same antibiotic treatment as the previous year. The other six individuals did not take any antibiotics before the flu shot on day four of the experiment.
Similarly to the first clinical trial, the microbiome population crashed in individuals treated with antibiotics. However, in all eleven volunteers, the blood at the injection site of the flu vaccine was not able to produce an essential antibody need to fight an influenza virus.
Besides the bacterial depletion, individuals in the second clinical trial experienced widespread inflammation, but particularly of the intestinal tract. This common symptom of vaccine administration usually affects people over 65. The body cannot make secondary bile acids because the production requires bacterial assistance in converting primary bile acids produced in the liver. The secondary bile acids are responsible for reducing inflammation, and without them, the GI tract cannot mitigate irritation caused by the vaccine.
These clinical trials make it clear that antibiotic affect the human immune system far more than previously known. The study provides more evidence that receiving a flu shot every year is helpful to the immune system, even if antibiotics have reduced the microbiome population.
Sources: Stanford Medicine, Cell