Harnessing the Power of Natural Killer Cells to Fight Cancer
Manipulating the immune system’s population of natural killer cells could bolster therapies targeting cancer. A new study saw positive results investigating the cells in mice, and scientists show that human cells share the same promising physiological responses.
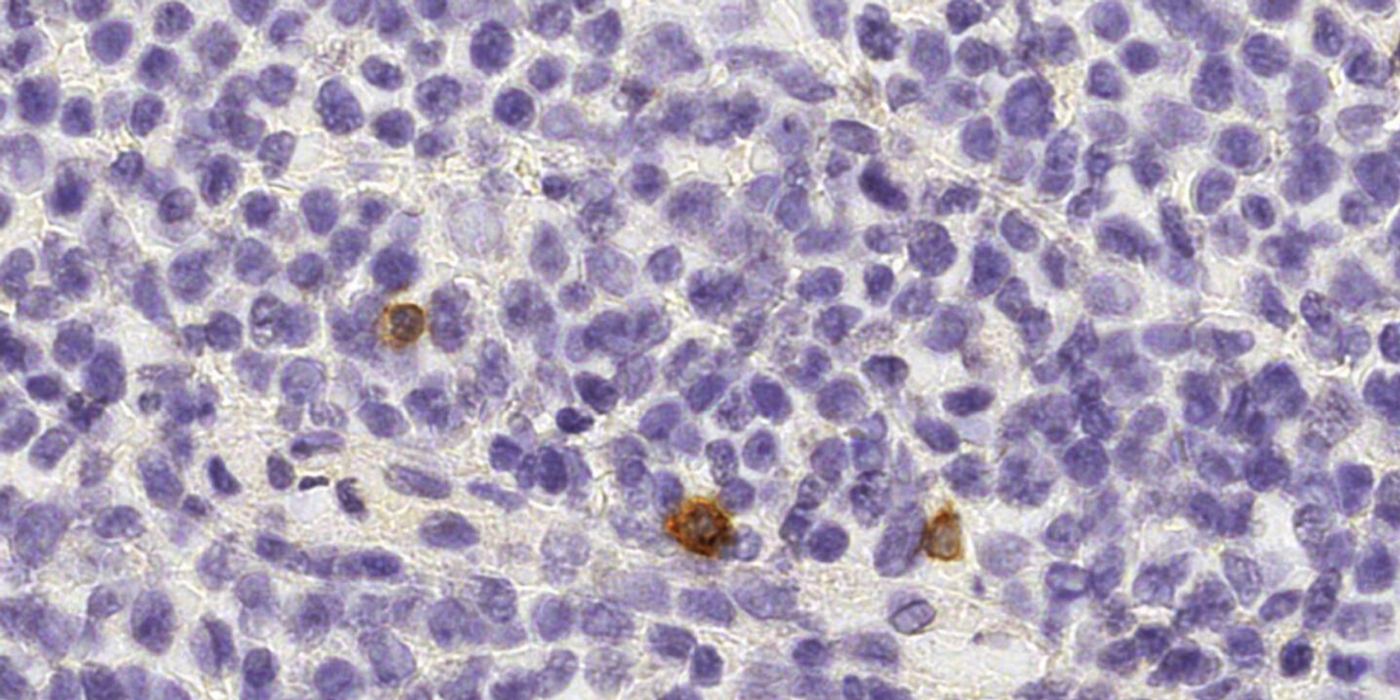
NK cells (brown) patrolling between cells of a healthy human lymph node. Source: Institute of Pathology, University of Bern
Natural killer (NK) cells are key components of the innate immune system, the arm of human immunity that responds first to pathogenic invasions with a non-specific attack. That attack could be via virus or cancer, but either way, NK cells seek to eliminate affected cells before the affliction has a chance to spread. They can do this one of two ways: release cell toxins for a direct kill, or activate cells affected by cancer or virus to self-destruct, a process called apoptosis.
During a viral invasion, NK cells manage T cells of the adaptive immune system, the second, more specific phase of immune response, by preventing them from unnecessarily killing host cells unaffected by the virus. NK cells do this by releasing “messenger molecules” to support immune defense.
But scientists from the recent study are presently more interested in NK cell-induced cellular self-destruction and its potential ability to improve anti-cancer clinical tactics. Researchers put “death ligands” under the microscope, scrutinizing these surface proteins’ abilities of interacting with “corresponding receptors” on the surface of target cells. One death ligand stood out during the study, a protein called “tumor necrosis factor-related apoptosis-inducing ligand,” or “TRAIL.”
Cancerous cells are more sensitive to TRAIL stimulation than healthy cells, making TRAIL-related therapies potential options for initiating the apoptotic self-destruction of cancer cells. One roadblock seems to stand in the way: in past studies, TRAIL has also been associated with negative health outcomes in particular infection cases. The present study offers a solution for navigating around this roadblock to effectively utilize TRAIL as an anti-cancer tactic.
Researchers began by studying the TRAIL response in a murine model of viral infection. Mice whose NK cells did not have TRAIL were better equipped to fight the virus than mice whose TRAIL was fully intact. NK cells perceived cellular signaling differently when they lacked TRAIL; they did less killing and more messenger molecule production, and T cells subsequently became more protective and were able to better remove virally infected cells. Researchers saw signs of similar activity in human NK cells too, indicating a potential for the reprogramming of NK cells to better target cancerous cells in addition to virally infected cells.
Sources: University of Bern, European Molecular Biology Organization (EMBO) Reports