B Cell Surface Features Revealed by Advanced Microscopy
With machine learning methods and an advanced technique called lattice light sheet microscopy (LLSM), scientists have now revealed structures on the surface of B cells that influence how those immune cells are distributed. The study authors suggested the function of antigen receptors on those immune cells may also be affected by those surface structures. The findings have been reported in The EMBO Journal.
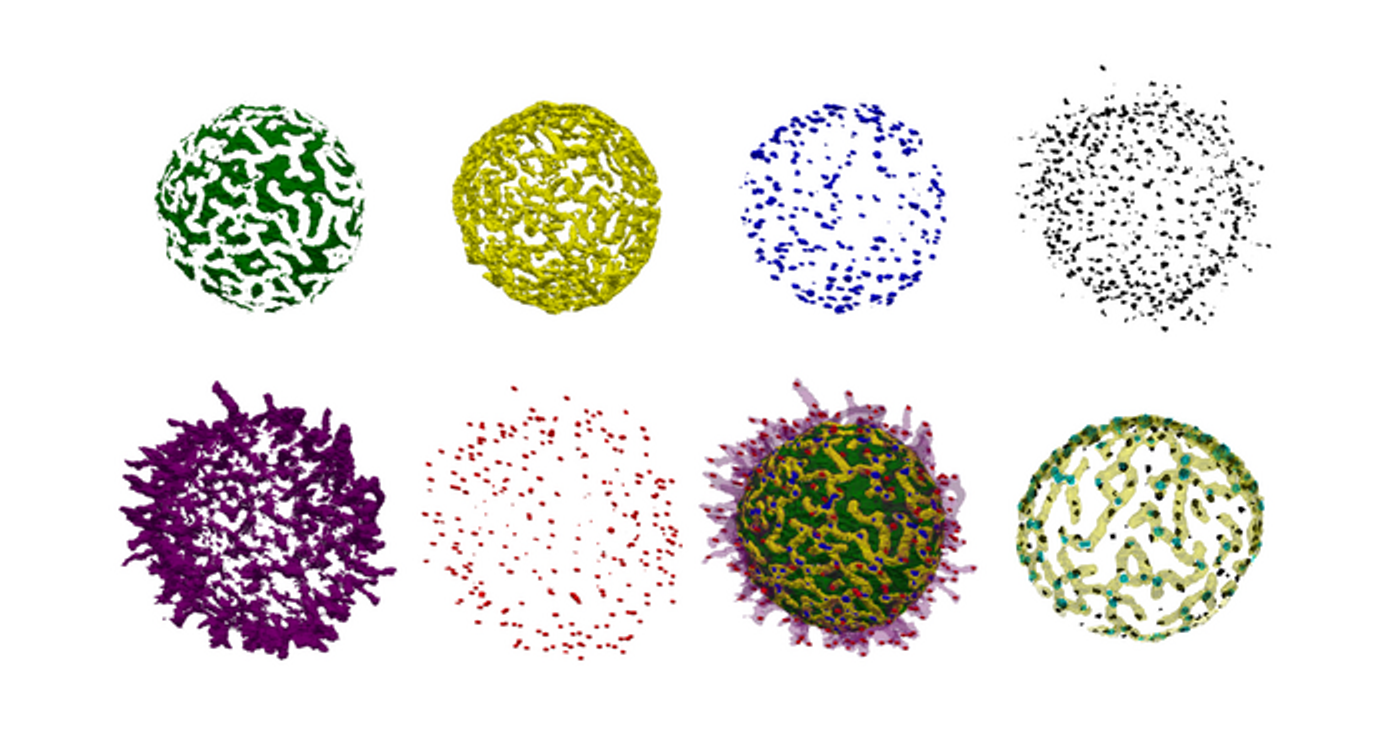
The antigens on the surface of B cells help them identify pathogens. Researchers investigated the distribution of those antigens on live cells, and found that B cell surfaces feature a landscape of ridges and protrusions that are linked together. A type of antigen receptor called IgM-BCRs were found to accumulate in certain places. The location and clustering of those antigens into larger structures may influence antigen activity and consequently, how B cells are activated.
B cells do not have smooth surfaces with receptors distributed at seemingly random locations, as they are often depicted in textbook illustrations. Instead, they have structures called microvilli protruding from the surface. These microvilli help the cell look for the antigens that are on the surfaces of pathogens. B cells recognize pathogenic antigens with different types of B cell antigen receptors (BCRs). When antigen receptors are activated, they can trigger a cascade of biochemical signals that leads to the production of antibodies.
In this work, the researchers used LLSM to image the surface of live B cells in three dimensions. Instead of being on a flat surface, which can change the outer structure of a B cell, the researchers observed them in a 3D culture that mimics live tissue and is more representative of their normal environment, noted first study author Dr. Deniz Saltukoglu from Freiburg University. LLSM images the cells at high speed, and without causing major disturbances.
The team developed a special analysis tool to analyze the data they generated from the LLSM, drawing inspirations from algorithms that have been applied to geographical data from archeological surveys. A cellular landscape was generated, and the researchers saw that IgM-BCRs cluster together, mainly along the ridges, and close to the bases of microvilli. The movement of ridges on the cell surface was also linked to the clusters' positions.
“We think that the 3D location of the antigen receptors controls their activity,” said Prof. Dr. Michael Reth of the Clusters of Excellence BIOSS and CIBSS (Centre for Integrative Biological Signaling Studies) at the University of Freiburg. “Localization at the microvilli base may prevent their unwanted activation. Once B cells receive a danger signal, they [extend] their microvilli and we assume that the IgM-BCR clusters then get recruited to the tip where they are localized in an optimal position for antigen sensing.”
Sources: University of Freiburg, The EMBO Journal