Resolving Immune Confliction to Battle Cancer
Mice models are more often than not the models of disease used to study the interaction between immune cells and cancer. A new study looks closer at whether or not mice models are as similar to human cells as some studies suggest. Plus, researchers investigate myeloid-derived suppressor cells (MDSCs) and their involvement in cancer progression.
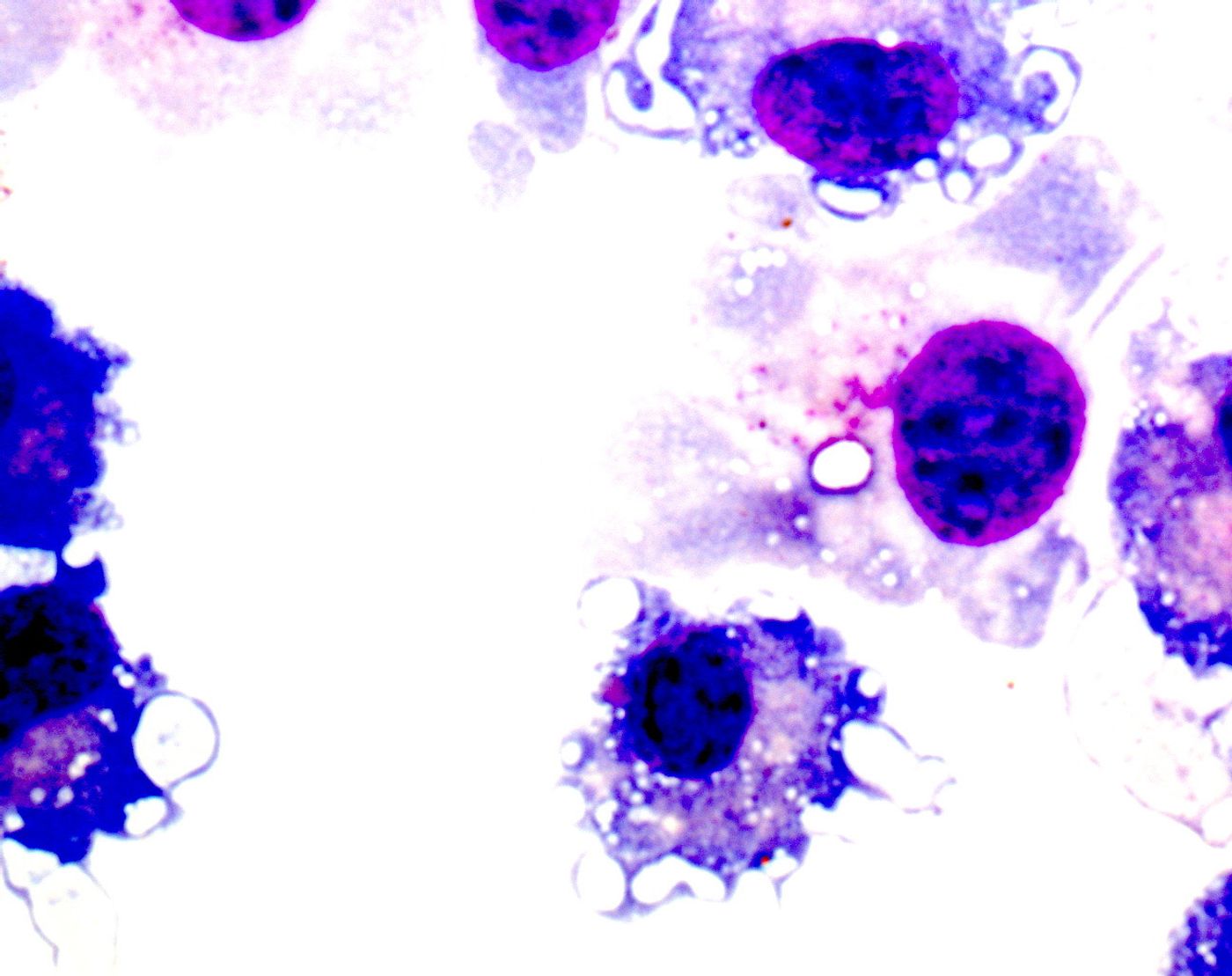
Myeloid-derived suppressor cells help the immune system to relax, as opposed to other immune cells in the body that are designed to get the immune system revved up. Therefore, it makes sense that tumors might be interested in increasing the number of MDSCs to prevent a full-blown immune attack.
Before beginning experiments to learn more about MDSC regulation and cancer, the researchers paused to ask, why mice? “Many versions of vaccines and tumor models rely on the responses of [spleen white blood cells from mice],” explained study investigator Martin McCarter, MD. “But it turns out we don't know as much about human [spleen white blood cells] and their immunologic importance. There's a big leap of faith that mouse models are applicable to humans.”
McCarter and a team of researchers from the University of Colorado Anschutz Medical Campus conducted a study looking at immune cells from the spleens of 26 patients with different types of cancer, looking for similarities and differences between human and mouse cells in the spleen.
They found that mouse spleen white blood cells are plentiful and easy to isolate, while the supply of human counterpart cells is lacking and these cells are difficult to isolate due to their complex mix of markers. "Now with this paper we show how future researchers can isolate these human [spleen cells], hopefully leading to more work in this area,’ said first author Kim Jordan, PhD.
Next, in a comparison of cancer patient spleen cells to cells from patients with benign pancreatic cysts, researchers saw that in both mice and humans, immune cells in the spleen are more numerous in cancer patients than in non-cancer patients. They believe that this association suggests that immunosuppressive spleen cells that block T cell responses may be contributing to tumor growth.
Their immunosuppressive behavior is a regulatory function that is beneficial in the realm of stopping adverse autoimmune reactions but harmful in the case of cancer, where the immune system can use all the help it can get. The University of Colorado study showed that the more immunosuppressive cells there were in the spleen, the more the cancer patients suffered.
Treatment options exist to boost the immune system during cancer. For example, there are immunotherapies that re-induce T cells to fight cancer cells, PD-1 and PD-L1 inhibitors, which activate T cells by preventing tumor cells from suppressing them. Also, CAR-T cell therapy enables T cells to differentiate between human host cells and tumor cells so they know which type of cell from which they should take direction. Despite these existing T cell-based treatments, cancer cells can still inhibit the immune system by enhancing the production of MDSCs.
“There are many elements of the immune system we don't understand, for example how tumors manipulate or utilize a patient's own immune system to block the immune response against their own tissue,” McCarter said. “Now we are taking steps to understand this process, and understanding the basic science allows us the opportunity to intervene with therapies to stop it.”
Now with a better understanding of both the similarities and differences between human cells and mice cells as well as the prevalence of spleen immune cells in cancer patients, University of Colorado scientists plan on human clinical trials that target MDSCs as a supplemental treatment to existing therapies.
The present study was published in the journal Cancer Immunology & Immunotherapy.