Predicting Melanoma Patients' Responses to Immunotherapy
There’s a new way to predict if cancer patients will respond well to newly FDA-approved immunotherapy drugs for cancer. From the University of California, San Francisco, researchers take a look at melanoma patients and the power of the new predictive biomarker.
Immunotherapy is sometimes applied as a combination of two drugs with different targets. Past studies showed UCSF scientists that whether a cancer patient needs combination therapy or not depends on the number of immune cells in the tumor. More immune cells, the more likely a cancer patient is to benefit from just one immunotherapy drug.
Why would a person respond to one better than the other?
"Combination immunotherapy is super-expensive and very toxic," explained senior author Adil Daud, MD. "You're putting patients at a lot of extra risk if they don't need it, and you can adjust for that risk by knowing in advance who can benefit."
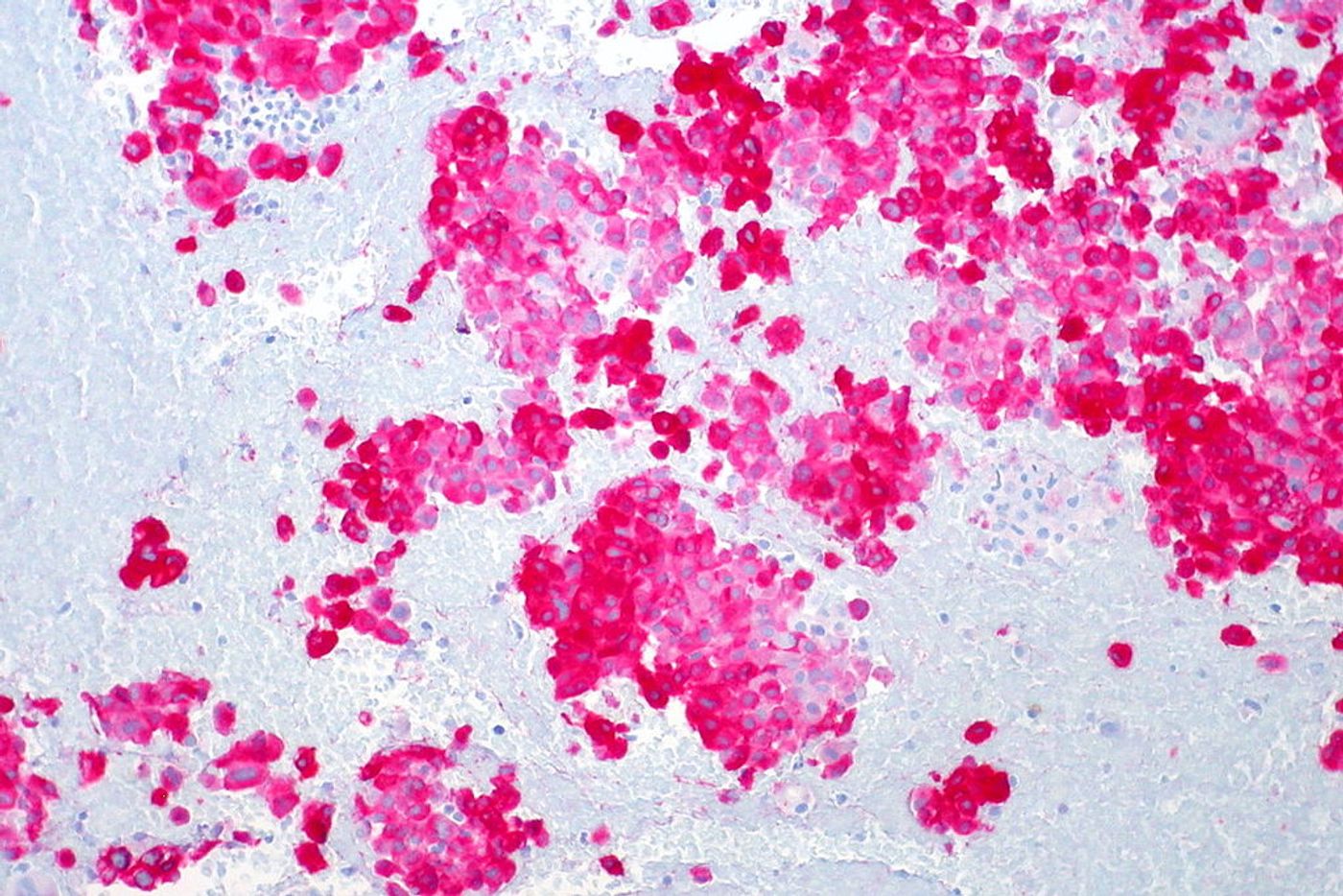
The new assay measures the amount of T cells in tumors so medical professionals can predict how an individual would respond to immunotherapy. T cells work in the immune system to recognize certain proteins on pathogens and cancer cells that distinguishes them from healthy human cells. However, cancer cells hide from the immune system by masquerading as healthy cells, wearing the same protein, PD-L1, on their surface that T cells see and ignore them. Cancer cells then escape undetected, and tumors continue to grow inhibited by the immune system.
Immunotherapy checkpoint inhibitor drugs block either PD-L1 or PD-1, the protein on the surface of T cells that helps them recognize PD-L1. These drugs make cancer visible to the immune system again. But, existing FDA-approved checkpoint inhibitors work for only 20 to 40 percent of cancer patients.
The new UCSF study took tumor samples from 102 melanoma patients and used sorting equipment to calculate the number of T cells in the tumor samples.
"You're pushing on two different gas pedals - PD-1 and CTLA-4," Daud said. CTLA-4 is another checkpoint protein targeted by immunotherapy drugs. "If you're one of those patients with a low number of exhausted T cells, you have a better likelihood of benefitting from both drugs."
During their study, they also saw that women in particular were more likely to benefit from combination immunotherapy. They are also hoping to extend the predictive abilities of the assay to other types of tumors.
The present study was published in the Journal of Clinical Investigation Insight.
Source: UCSF