Explosive Cell Death Starts A "Fire" in the Lungs
An “explosive” form of cell death, called pyroptosis, is part of the normal immune response to the toxins released by bacteria during an infection. But when the infection reaches the lungs, pyroptosis does more harm than good.
From the University of Illinois at Chicago, researchers investigate what happens at the cellular level when bacteria invade the body and release a toxin called lipopolysaccharide (LPS). This is a toxin produced by a specific group of bacteria that stimulates innate immunity in all kinds of organisms, including humans.
In the present study, researchers conducted experiments in mice, where an enzyme called caspase-11 initiates pyroptosis in response to LPS. But the process is similar in humans, where caspases 4 and 5 “start the fire.”
“In pyroptosis, cells go out with a bang,” said co-lead author Dr. Jalees Rehman.
Caspases activate pro-inflammatory cytokines, immune messengers that convey a variety of messages. In response to these particular cytokines, the cell membrane breaks down, releasing a flood of cytokines that send warning signals to neighboring cells communicating the presence of the bacteria.
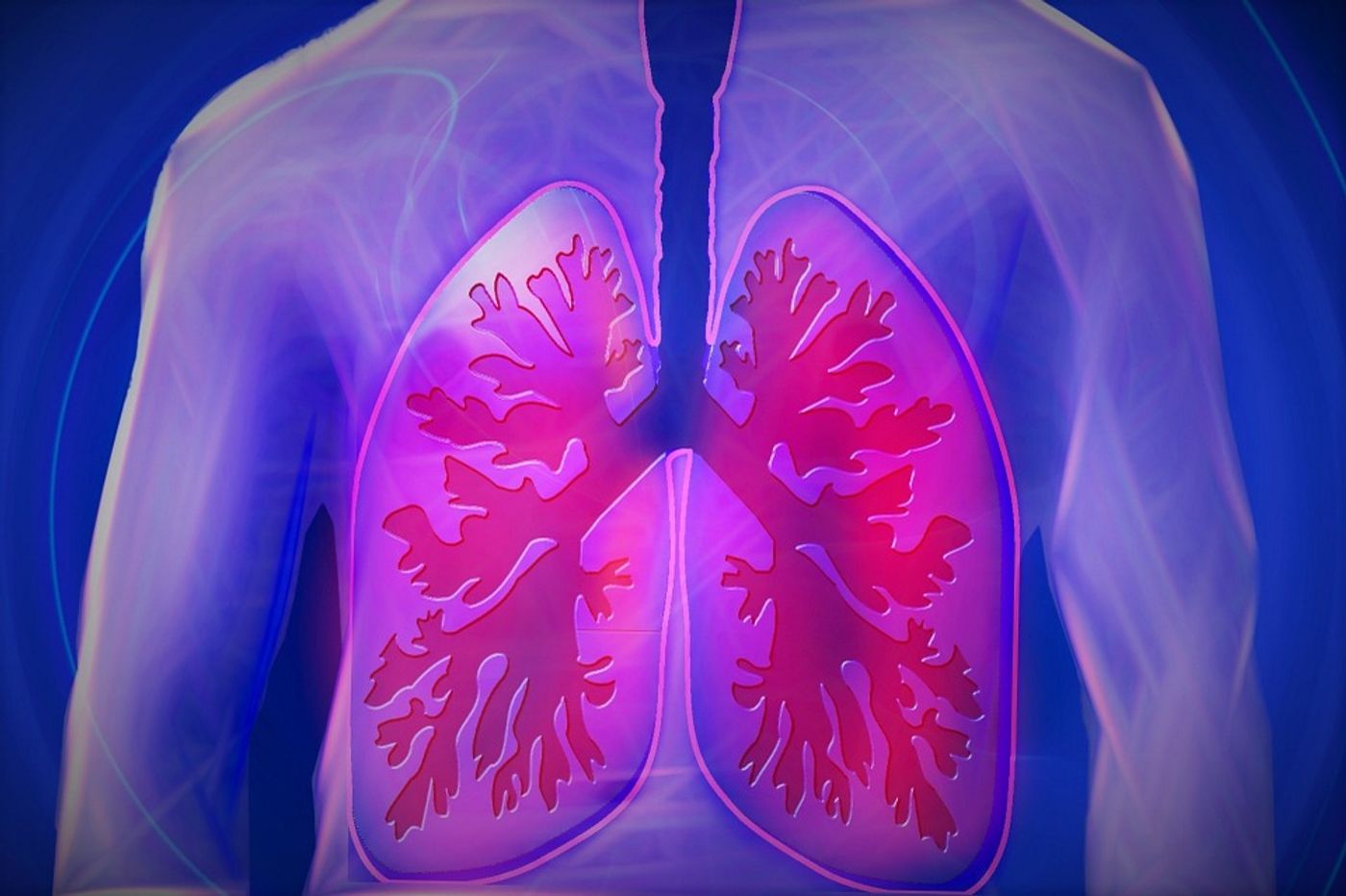
Sometimes bacteria enter the bloodstream, opening up the possibility of the infection reaching blood vessels in the lungs. The response of cells lining the blood vessels, known as endothelial cells, to LPS is bad; the endothelium becomes “leaky,” granting lung access to fluid it normally restricts from entering. This leads to acute lung injury.
Endothelial cells are uniquely able to adjust their number and arrangement to accommodate the local tissue. This ability is largely what enables them to promote tissue growth and repair. But when a bacterial infection affects endothelial cells, lung tissue is put in jeopardy, with gas exchange not functioning at its full potential. Acute lung injury has a high death rate, resulting in 30 to 40 percent of the 200,000 people affected by the condition in the United States every year to die. Treatment options for acute lung injury mostly include artificial ventilation while the infection is being treated with antibiotics.
In the University of Illinois at Chicago study, researchers used mice models injected with LPS to observe how infections can lead to acute lung injury. As expected, they saw high mortality rates when the infection reached the endothelial cells. However, when researchers manually erased all traces of caspase 11 in the endothelial cells of the lung blood vessels, more mice survived and endothelial cells let in less fluid to the lungs
"Caspase-11 is clearly required for the activation of pyroptosis in the endothelial cells of mice," Rehman said. "Drugs that block this pathway or the corresponding human caspases 4 and 5 could help prevent or reduce the extent of lung injury."
Rehman and other scientists are convinced that the cellular response to LPS evolved as part of an “alarm signaling process” to wake up the immune system, promote inflammation via cytokines, and attack the invading bacteria. Although the response may be “well-intended” by nature, this present study shows scientists that they may need new targets for drugs to prevent infection-induced tissue damage.
The present study was published in the Journal of Clinical Investigation.
Sources: Journal of Endotoxin Research, Molecular Biology of the Cell 4th edition, University of Illinois at Chicago