"Runx3" Protein Directs Killer Cells to Set Up Camp in Tumors
A certain type of T cells from the immune system is responsible for infiltrating cells infected by bacteria and viruses as well as cancerous cells that grow uncontrollably. These cells grow in numbers in the spleen and lymph nodes, but what signal indicates to the T cells that it’s time to move, either to infected tissues or to growing tumors?
From the Scripps Research Institute, in a study published in Nature, scientists explore the movement of CD8+ T cells, or killer T cells.
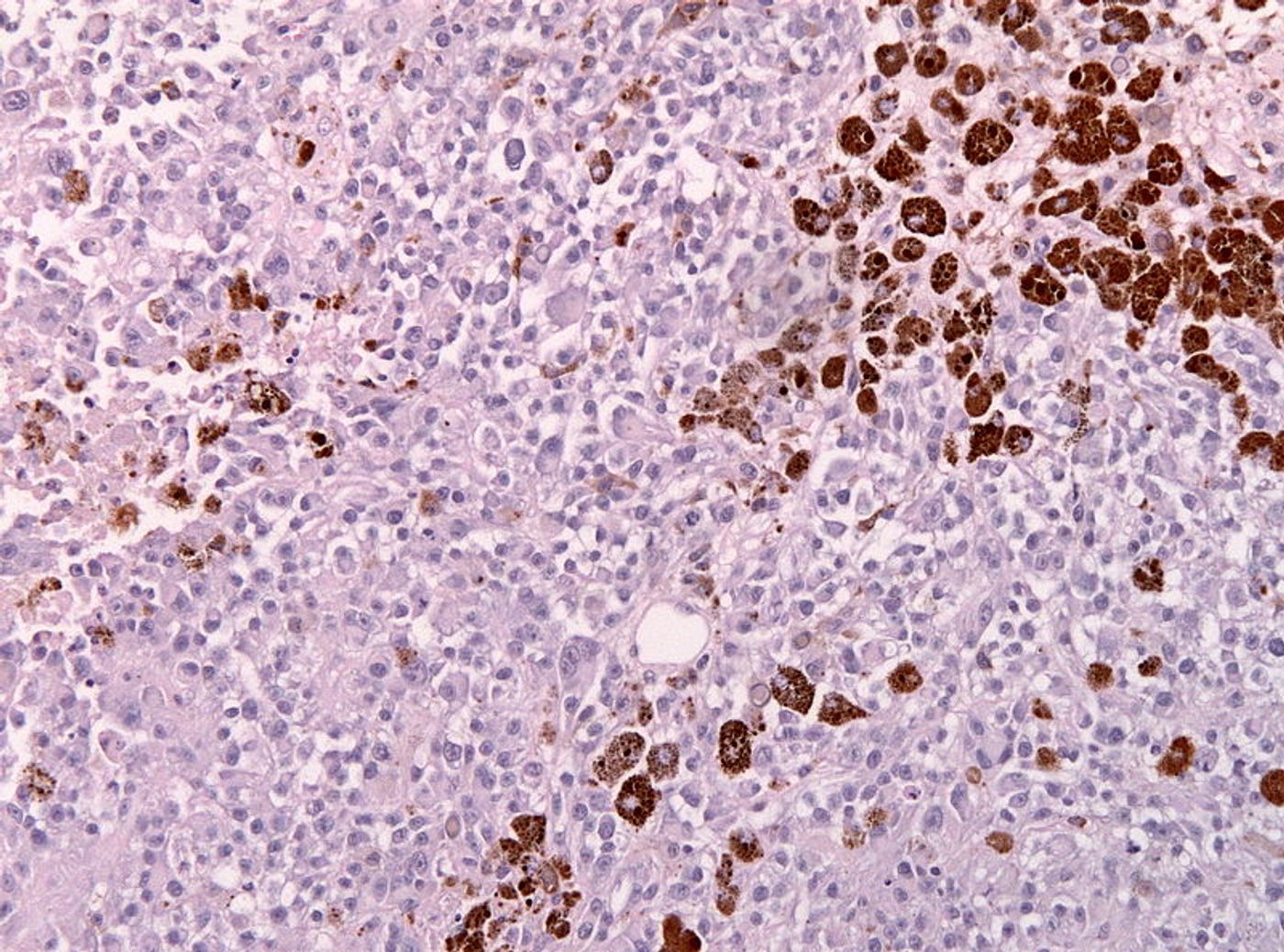
The study began with the discovery of a protein called “Runx3,” which directs the movement of killer T cells, from the lymphoid system to tumors and infected tissues. Runx3 does this by interacting with chromosomes inside killer T cells, programming genes in such a way that killer T cells congregate in a solid tumor.
Thus, Runx3 could be useful in certain killer T cell-based cancer immunotherapies such as checkpoint inhibitor blockages, which promotes the accumulation of killer T cells in tumors, and adoptive cell transfer, which extracts, refines, and returns a cancer patient’s T cells to mount a specific and strengthened attack against a certain type of cancer.
In the past, adoptive cell transfer immunotherapy approaches were successful against blood cancer, not so much for solid tumors. But, improving the communication between killer T cells and Runx3 could potentially improve the anti-cancer efforts of the immune system.
“We could probably use Runx3 to reprogram adoptively transferred cells to help drive them to amass in solid tumors," explained Matthew Pipkin, PhD.
Pipkin’s study was a comparison of killer T cell gene expression from non-lymphoid tissue to gene expression from T cells in the circulation. Researchers found a “distinct pattern.”
"The screens showed that Runx3 is one at the top of a list of regulators essential for T cells to reside in nonlymphoid tissues,” Pipkin explained. Runx3 made certain genetic changes in killer T cells to draw them to tissues and tumors, genetic changes found in killer T cells that naturally reside in these places.
Next researchers designed killer T cells genetically altered to overexpress Runx3 and used them in adoptive cell transfer in mouse models of melanoma. These cells effectively inhibited cancer growth, and mice who received this treatment lived significantly longer compared to untreated mice.
Sources: Scripps Research Institute, British Society for Immunology