Successful Neural Stem Cell Therapy Without Immunosuppression
Neural stem cells designed to be identical to the host they were derived from are the newest hope for scientists in transplantation medicine, specifically for restoring tissues damaged by a spinal cord injury. From the University of California - San Diego, scientists show how they successfully transplanted such cells into pigs, which are similar to humans in the context of the central nervous system.
Designing custom neural stem cells from genetically different donors can be difficult. Like any transplantation, inserting foreign cells carries a risk of the host immune system attacking the cells, causing a system-wide inflammatory response.
Giving a transplant patient immunosuppressive drugs prior to the transplantation may reduce the risk of tissue rejection, but the prescription also delivers all the risks that come with suppressing the immune system: leaving the body vulnerable to infections from bacteria, viruses, and parasites.
In their new study, UCSD scientists successfully designed neural precursor cells from induced pluripotent stem cells (iPSCs). Senior author Martin Marsala, MD, lauds human iPSCs as the “ultimate source of cells to be used in future clinical trials for treatment of spinal cord and central nervous system injuries in a syngeneic or allogeneic setting.”
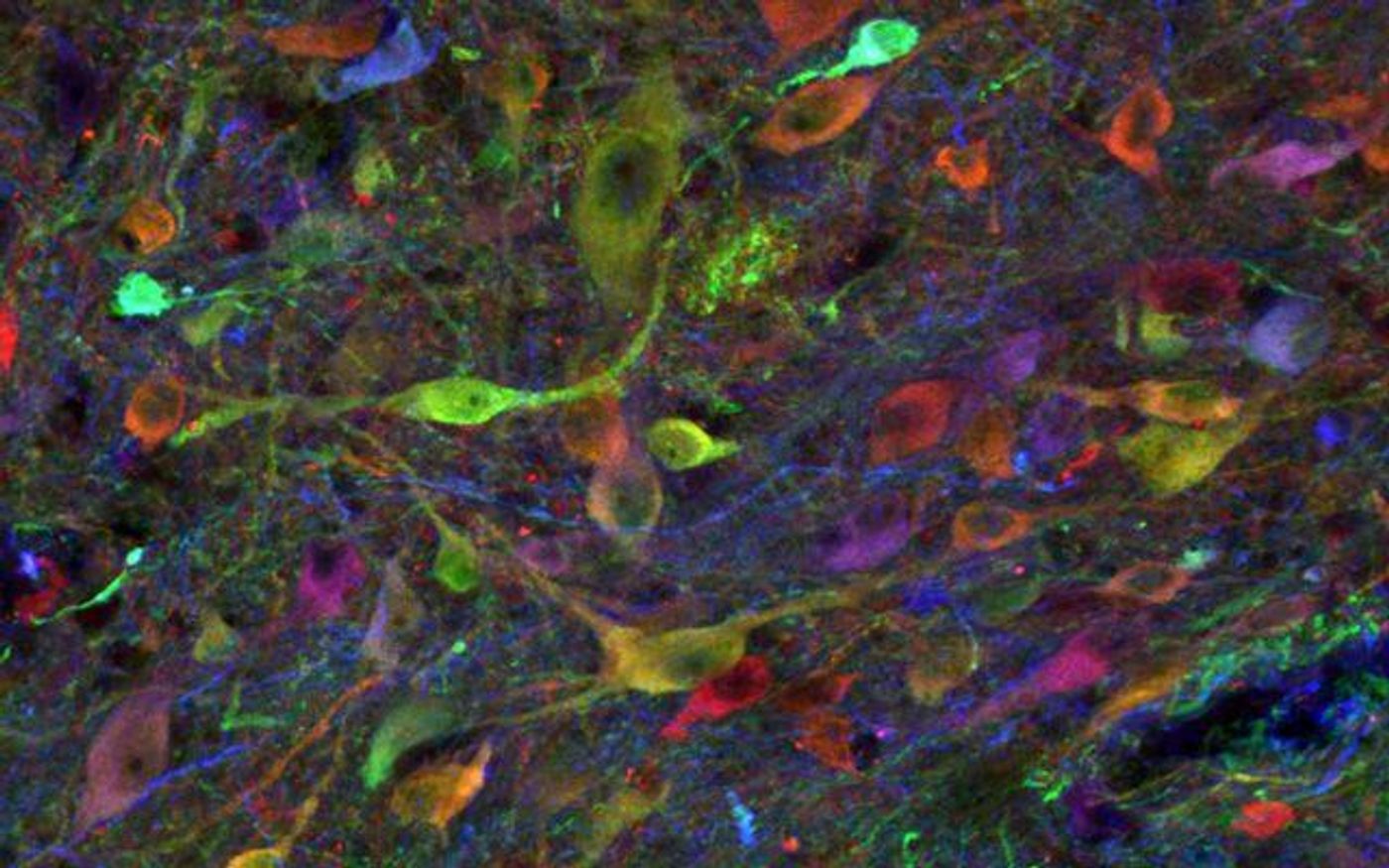
Researchers implanted the neural cells into the spinal cords of genetically identical adult pigs without applying any immunosuppressive drugs, and they observed long-term, cancer-free survival. Not only did the cells survive, but they also differentiated into neurons and supporting glial cells needed to rebuild damaged tissue.
Marsala and the others saw similar positive results in adult pigs with different genetic backgrounds. These pigs also had chronic spinal cord injuries, unlike the genetically identical pigs who were not injured.
"Using RNA sequencing and innovative bioinformatic methods to deconvolute the RNA's species-of-origin, the research team demonstrated that pig iPSC-derived neural precursors safely acquire the genetic characteristics of mature CNS tissue even after transplantation into rat brains,” explained co-author Samuel Pfaff, PhD.
"We took skin cells from an adult pig, an animal species with strong similarities to humans in spinal cord and central nervous system anatomy and function, reprogrammed them back to stem cells, then induced them to become neural precursor cells (NPCs), destined to become nerve cells,” Marsala explained. “Because they are syngeneic - genetically identical with the cell-graft recipient pig - they are immunologically compatible. They grow and differentiate with no immunosuppression required."
The present study was published in the journal Science Translational Medicine.