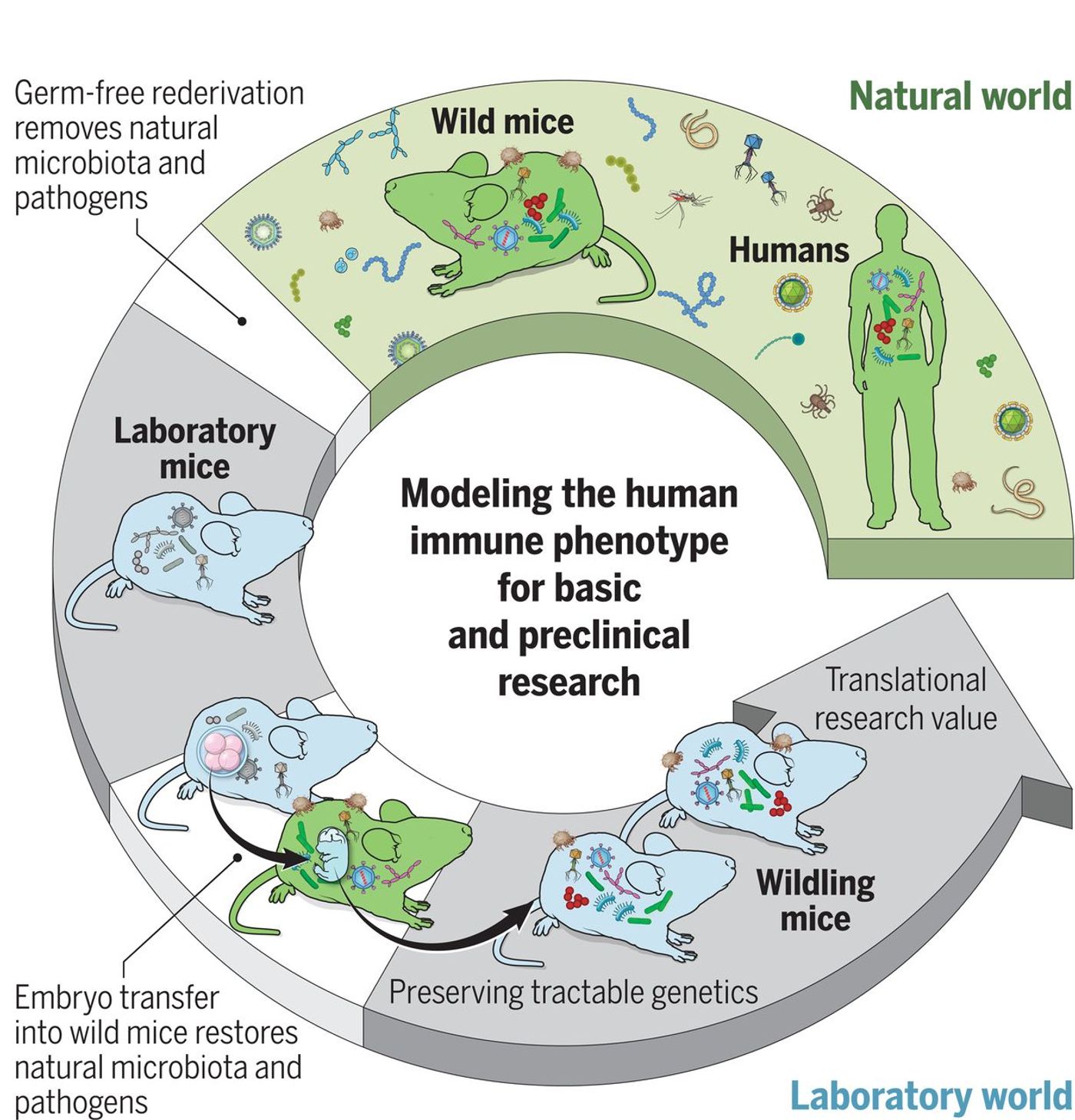
Lab Mice Born to Moms From the Wild Make Better Research Models
Researchers have used mouse models for decades. While they have been the source of some important discoveries, there has been some criticism of mouse studies that may be poorly designed or don’t mimic human conditions. Some research has been difficult to reproduce, calling conclusions into question. Studies have also suggested that the environment of lab mice is so far from what's experienced in the natural world that they cannot faithfully replicate a free-living mammal’s physiology. One reason why wild animals are so physiologically different from their laboratory counterparts is the microbiome.
The microbes that we and many other animals carry, especially those in our gastrointestinal tract - also known as the gut microbiome, have been shown to have a strong influence on the host’s health and well-being. Imbalances in the microbiome have been connected to a variety of human diseases. Some of that work has been conducted using human samples, but other studies have used research animals.
Mice used in research are classified by strain and there is often a wealth of physiological data available about them, especially one strain in particular, C57BL/6, which is a mouse that can be commonly found in the lab. Importantly, there is a high-quality reference genomic sequence for this mouse.
A new study has found that by implanting C57BL/6 embryos into mice from the wild, the resulting colony, which the study authors called wildlings, were more like their wild counterparts. They still carried the genome we know so much about, but the physiology of the wilding mouse is more like a mammal that lives freely, and has an immune system that has evolved to withstand the natural world as opposed to the industrial environment of research facilities.
The researchers found that the wildling colony was different from typical lab mice when it came to the viruses, fungi, and bacteria they carried in their gut, as well as the microbiome of their skin and vagina. This wildling microbiome was found to be stable over generations and withstood exposure to antibiotics or diet variation.
In this study, the investigators evaluated their model, focusing on the immune system of wild and laboratory mice as well as the wildlings they created. After surveying several sites including the gut and skin, the researchers found that the immune system of the wildlings was most like their wild counterparts when it came to the characteristics observed in the blood and spleen.
The researchers wanted to check the validity of the wildling mouse model they created. They determined whether it is an improvement over current rodent models by focusing on old research conclusions that did not hold up once they were tested in the clinic on human patients. One such assay looked at the CD28-superagonist (CD28SA); in mice and other models, CD28SA had a therapeutic impact on autoimmune and inflammatory diseases, likely through the expansion of anti-inflammatory regulatory T cells (Tregs). In clinical trials, however, there were life-threatening immune side effects. In the wildling model, CD28SA did not expand Tregs and caused an immune response, contrary to the findings from lab mice.
The wildlings also served as a better model for testing the effects of anti-tumor necrosis factor-alpha (TNF-α) during septic shock. The treatment failed in people after working in animal models. The wildlings were also not rescued by this therapy, and acted as a better model of the human immune system in this case as well.
The researchers concluded that wildlings can be better for the study of a variety of diseases, including neurodegenerative and metabolic disorders. Using them may help improve reproducibility and translatability to the clinic.
Previous work has found that the lab mice don't carry a particularly healthy microbiome. It could be fortified, however, by adding gut microbes from wild mice to the guts of lab mice. The lab mice that received the wild-mouse microbes had better health. Those results are outlined in the video.
Source: Science