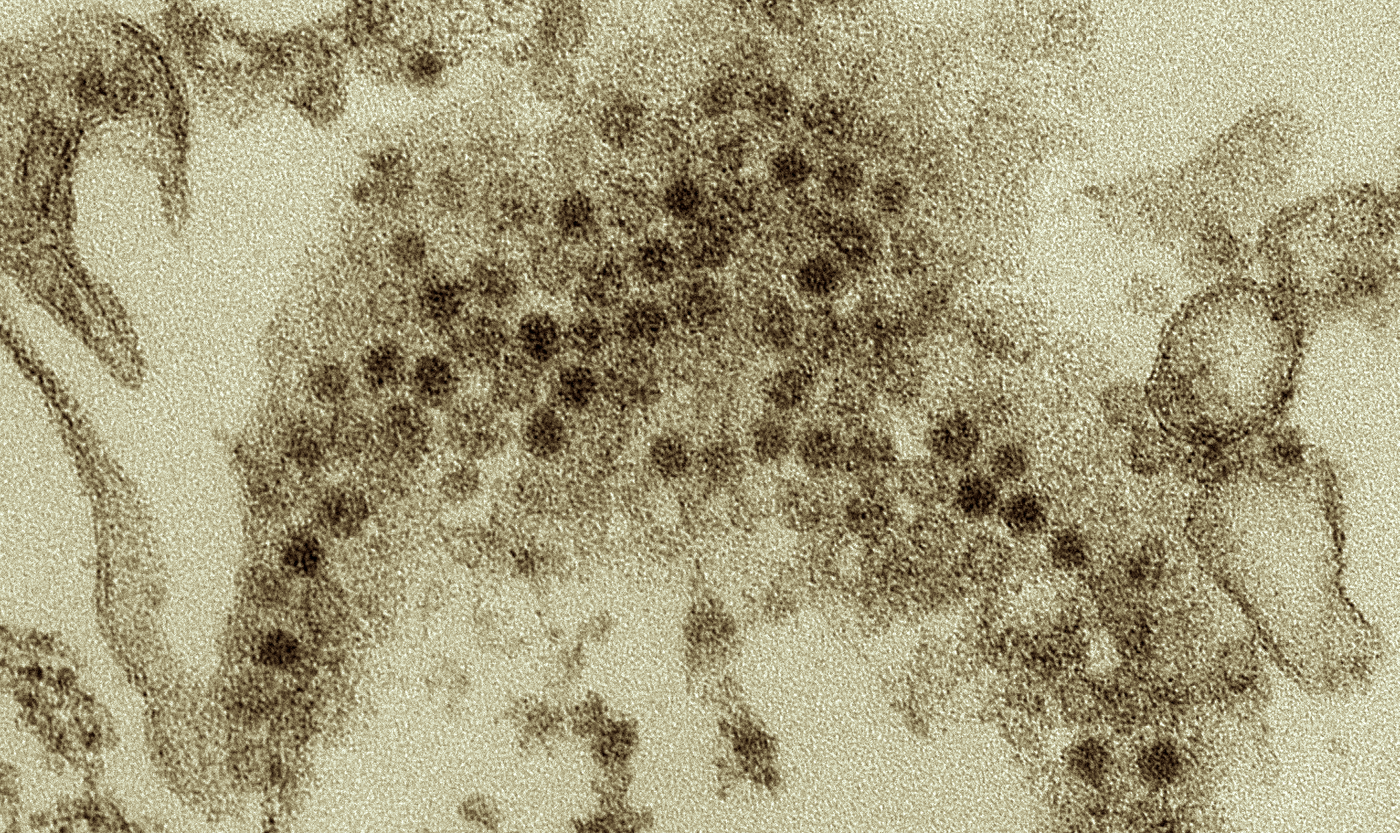
Why is Foot-and-Mouth Disease so Infectious?
There are two diseases with similar names: hand, foot, and mouth disease, which affects people, and foot-and-mouth disease, which affects cattle. Viruses from the Picornaviridae family cause these diseases, and they both result in painful foot and mouth blisters. They are also both extremely contagious (though the similarities apparently end there).
Scientists have now learned more about why the virus that causes foot-and-mouth disease, the version that affects cloven-hoofed animals, is so contagious. It affects animals in parts of South America, Asia, Africa, and the Middle East, and can spread when they move around; it's airborne. Poor farmers suffer the worst impacts, and losses are estimated to range from $6.5 billion to $22.5 billion every year.
Reporting in The FASEB Journal, an international team of researchers has studied a gene called 3B in the genome of the foot‐and‐mouth disease virus (FMDV); the genome contains three versions of the 3B sequence. This protein is also related to the replication of FMDV.
In this work, the researchers engineered viral fragments that carried one, or two, or all three of the various forms of 3B. They found that the fragments that carried all three forms were best at replication. They determined that having three forms of this protein has given FMDV a competitive advantage, which increases the chance it will survive and spread.
"Our experiments have shown that having three forms of 3B gives the virus an advantage and that probably plays a role in why the virus is so successful in infecting its hosts," said co-lead study author Dr. Oluwapelumi Adeyemi, formerly a researcher at the University of Leeds and now at the University of Ilorin.
"It is not as straightforward as saying because there are three forms of 3B - it is going to be three times as competitive. There is a more nuanced interplay going on which needs further investigation," added Adeyemi.
"The results of the data analysis were clear in that having multiple copies of the 3B protein gives the virus a competitive advantage. In terms of future research, the focus will be on why is that the case, and how the virus uses these multiple copies to its advantage," said co-lead study author Dr. Joe Ward, a post-doctoral researcher at Leeds. "If we can begin to answer that question, then there is a real possibility we will identify interventions that could control this virus."
Sources: AAAS/Eurekalert! via University of Leeds, Federation of American Societies for Experimental Biology