When Microbes Battle for Survival, the Weakest Can Win
Our world is filled with different types of bacteria, and they have to coexist with one another. They have to compete for space and resources in all kinds of environments, including inside the human body. Scientists are trying to learn more about how bacteria get along with one another. When they have to reside in one system, which bacterial species end up surviving and why?
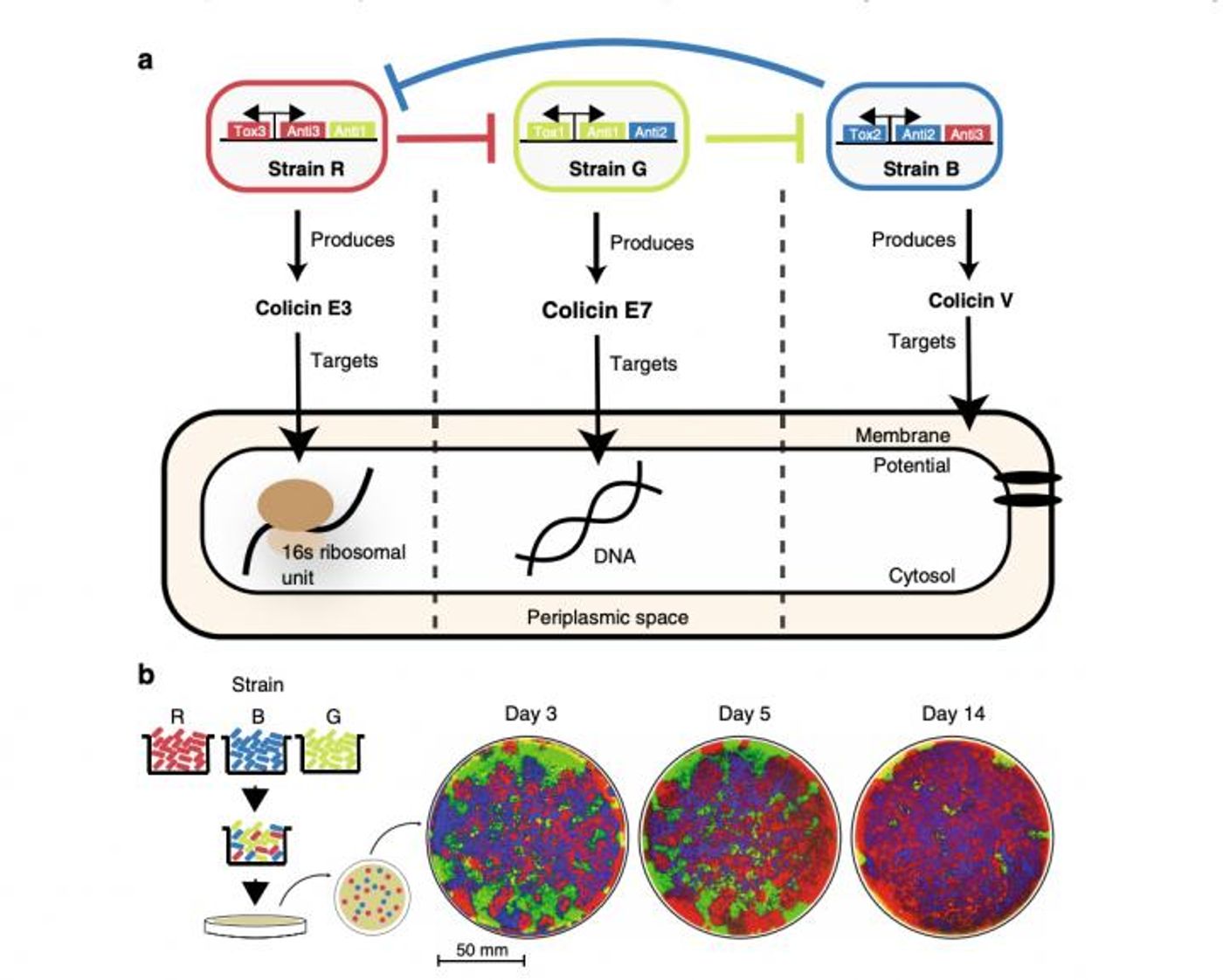
In a new study, researchers engineered three strains of Escherichia coli, which each generated a toxin that could kill one of the others. The three strains were then grown together, and the scientists found that it wasn't always the strongest strain, the one with the most powerful toxin that outlasted the others and was the bacterial survivor. The findings have been reported in Nature Communications.
"In synthetic biology, complex gene circuits are typically characterized in bacteria that are growing in well-mixed liquid cultures. However, many applications involve cells that are restricted to grow on a surface," noted Professor of Bioengineering and Molecular Biology Jeff Hasty. "We wanted to understand the behavior of small engineered ecologies when the interacting species are growing in an environment that is closer to how bacteria are likely to colonize the human body."
The three strains of E. coli were created and then mixed together on a Petri dish for a few weeks. The scientists found that the same microbial population would take over the whole surface of the dish time and time again, and it was not the microbe that made the most potent toxin. It had to be either the microbe that produced a toxin of 'medium' potency or the weakest one taking over the dish, and the researchers determined that it was actually the weaker population that conquered the other two.
It seems that the stronger E. coli strain can compete and win against the medium strain. That leaves the weaker strain without its medium enemy. It can then just slowly work against the stronger strain over time. The researchers devised mathematical models that can describe these relationships. It may be possible to use these findings to improve our understanding of the gut microbiome, and potentially help us learn more about engineering healthier ones.
Sources: AAAS/Eurekalert! via University of California - San Diego, Nature Communications