Some Bacteria Know the Time
People, animals, and even plants are known to have biological clocks, and new work has revealed that free-living bacteria do as well; even microbial cells are aligned with the planet's 24-hour cycle, and this alignment occurs in more than just photosynthetic microbes. While we've long known about the existence of these cycles in humans, we're still learning more about the circadian rhythms of different types of human cells and how they may cause disease when they are disrupted.
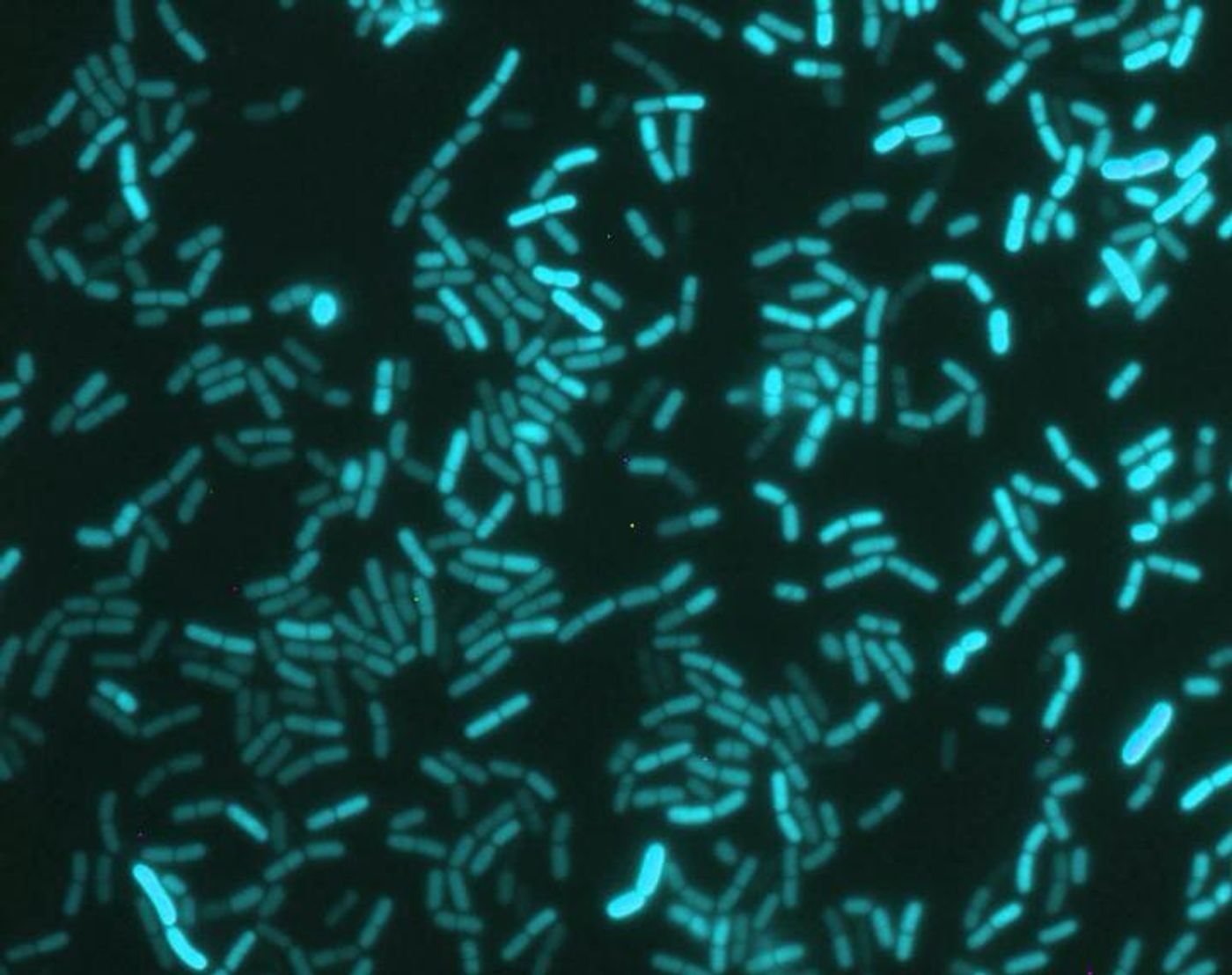
This study, which was reported in Science Advances focused on a microbe that does not photosynthesize light and lives in soil called Bacillus subtilis. Researchers used a tool called luciferase, which signals when a gene is active by lighting up. They wanted to know when two genes were active in total darkness compared to a cycle of twelve hours of dark and twelve of light. One gene under study, ytvA, encodes for a blue light photoreceptor and the other, KinC encodes for an enzyme that plays a role in biofilm and spore formation.
The research determined that the activity of ytvA was in sync with twelve-hour cycles of light and dark; the activity increased in darkness and decreased when it was light. The cycle remained even in constant darkness. It took several days for patterns of expression to emerge, and when the conditions were reversed, the pattern did as well. Circadian rhythms behave in a similar way.
"We've found for the first time that non-photosynthetic bacteria can tell the time. They adapt their molecular workings to the time of day by reading the cycles in the light or in the temperature environment," said lead study author Professor Martha Merrow, of Ludwig Maximilians University (LMU) Munich. "In addition to medical and ecological questions, we wish to use bacteria as a model system to understand circadian clock mechanisms. The lab tools for this bacterium are outstanding and should allow us to make rapid progress."
When other variables were tested, including temperature changes as Merrow noted, the scientists saw that the patterns of expression of ytvA and kinC changed like they did when light cycles were altered.
"Our study opens doors to investigate circadian rhythms across bacteria. Now that we have established that bacteria can tell the time we need to find out the processes that cause these rhythms to occur and understand why having a rhythm provides bacteria with an advantage," said study author Dr. Antony Dodd of the John Innes Centre.
"Bacillus subtilis is used in various applications from laundry detergent production to crop protection, besides recently exploiting as human and animal probiotics, thus engineering a biological clock in this bacterium will culminate in diverse biotechnological areas," added study co-author Professor Ákos Kovács of the Technical University of Denmark.
Sources: AAAS/Eurekalert! via John Innes Centre, Science Advances