How a Chronic Hepatitis B Infection Happens
Hepatitis B isn't usually a virus that causes immediately severe symptoms like Ebola or passes rapidly from person to person like measles, But hepatitis B still causes a serious illness that can become chronic, have lifelong consequences for patients, and which may be fatal for some. Researchers have now learned how five host cell proteins are involved in a critical step in the hepatitis B virus (HBV) life cycle. These findings, which were reported in Nature Communications, could help scientists develop a strategy for preventing these chronic infections.
"An estimated two billion people have been exposed to HBV, of whom 250 to 400 million are chronically infected," said senior study author Alexander Ploss, associate professor of molecular biology at Princeton University. "Currently, there is no cure for chronic HBV infection, and patients need to be on a lifelong antiviral regimen. Approximately 887,000 individuals die each year from HBV-related liver diseases or liver cancer."
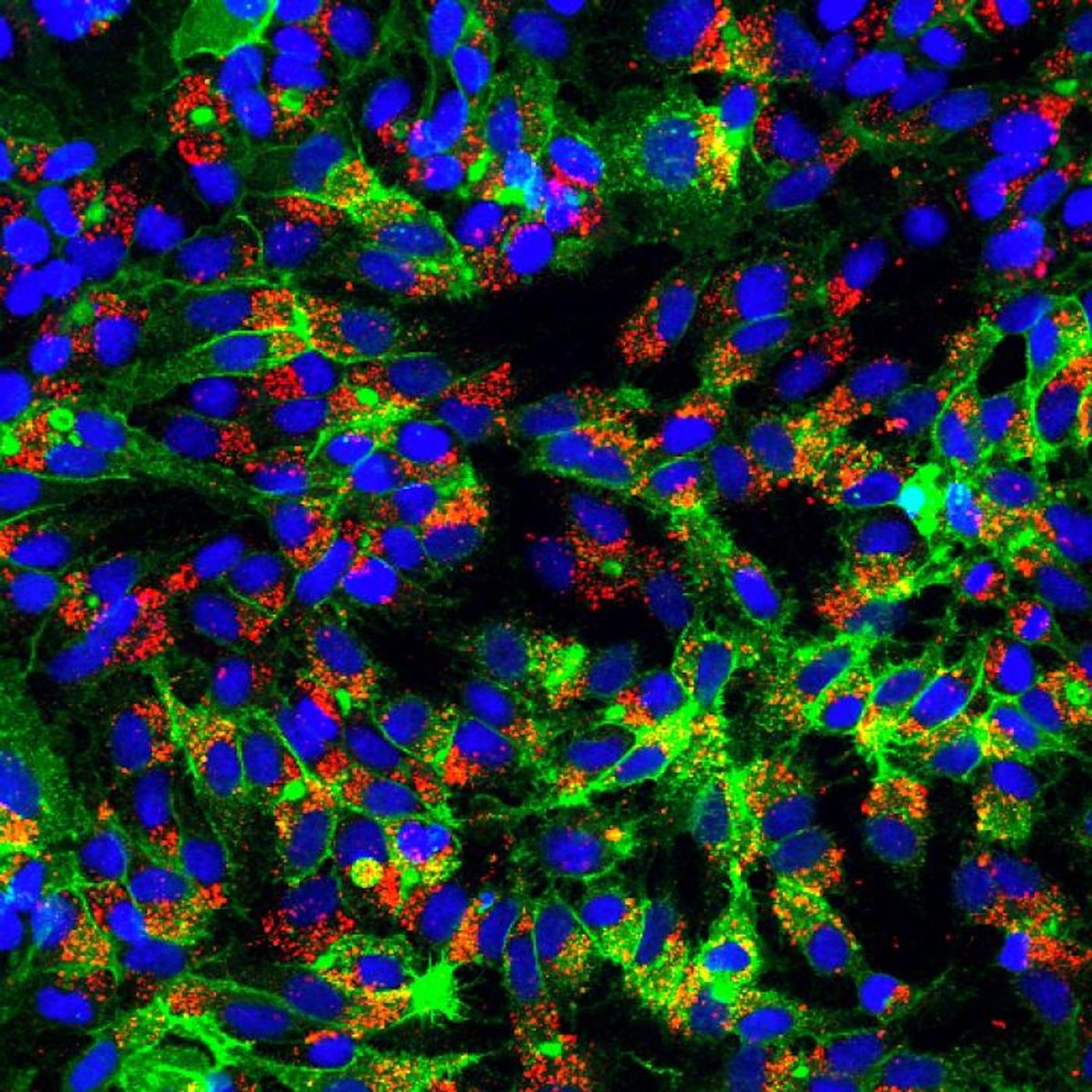
Ploss and colleagues have been working to reveal the details of HBV's life cycle. They've found that the viral DNA forms a structure that's essential to viral replication, and this conversion uses five proteins in the cell it infects to change into this structural form.
"Central to HBV replication is the formation of covalently closed circular DNA (cccDNA) from relaxed circular DNA (rcDNA) which is carried into the host cell by the virus during the initial infection," explained Ploss. "We have recently demonstrated that HBV relies on five host proteins - namely PCNA, RFC complex, POLδ, FEN-1, and LIG1 that are necessary and sufficient for this conversion step."
A DNA molecule is two strands of nucleotide bases that are complementary and bound to one another. The HBV genome is only four genes, which is extremely small. The virus has to rely on proteins in the host cell it infects to generate more viral particles, and to convert from rcDNA to cccDNA.
In the HBV rcDNA, the two strands have a gap in the sequence. On one strand the gap is considerably bigger, and it's offset from the other. Since these gaps in DNA are flagged as damage that needs repair by a cell (the cell's DNA repair machinery doesn't differentiate between this viral DNA and human DNA), they are filled in to create a circle of intact, double-stranded DNA. That's the cccDNA that hangs around in the nucleus of cells.
In this work, the researchers determined how this repair takes place and what proteins are involved. All five of the proteins play a role in fixing the DNA; two of the five proteins do repair work on one side, and all five work on the other.
"In this paper, [postdoctoral fellow Lei] Wei and Ploss provide a compelling story in elucidating the cellular machinery that is essential for converting the incoming HBV genome into the cccDNA," said HBV expert Dr. T. Jake Liang, a National Institutes of Health Distinguished Investigator in the Liver Diseases Branch. "This work offers not only important insights into the biochemical pathway of cccDNA biogenesis but also potential strategies to target cccDNA for therapeutic development."
The researchers have already determined that two compounds can interfere with the conversion of rcDNA to cccDNA in vitro. They are hoping that in the future, drugs like these compounds will be demonstrated to work in people.
"Our findings, biochemical approaches, and the novel reagents that we generated and engineered, open the door to providing an in-depth understanding [of] how this major human virus establishes persistence in host cells," Ploss said.
Sources: AAAS/Eurekalert! via Princeton University, Nature Communications