The gut microbiome, the complex community of bacteria carried in the gastrointestinal tract, is increasingly linked to health and disease in a range of ways. Researchers using live imaging in zebrafish have found that mechanical contractions in the gut have a major influence on bacterial competition in the gastrointestinal tract.
"The new findings could help improve such efforts and guide new treatments for diseases of the gastrointestinal system," explained Raghuveer Parthasarathy, a Professor of Physics and corresponding author of the new work. "This research shows that the physical environment and activity of the host intestine can be a major determinant of the bacterial makeup of the gut -- an idea that we think should very generally influence how we tackle issues of health and disease."
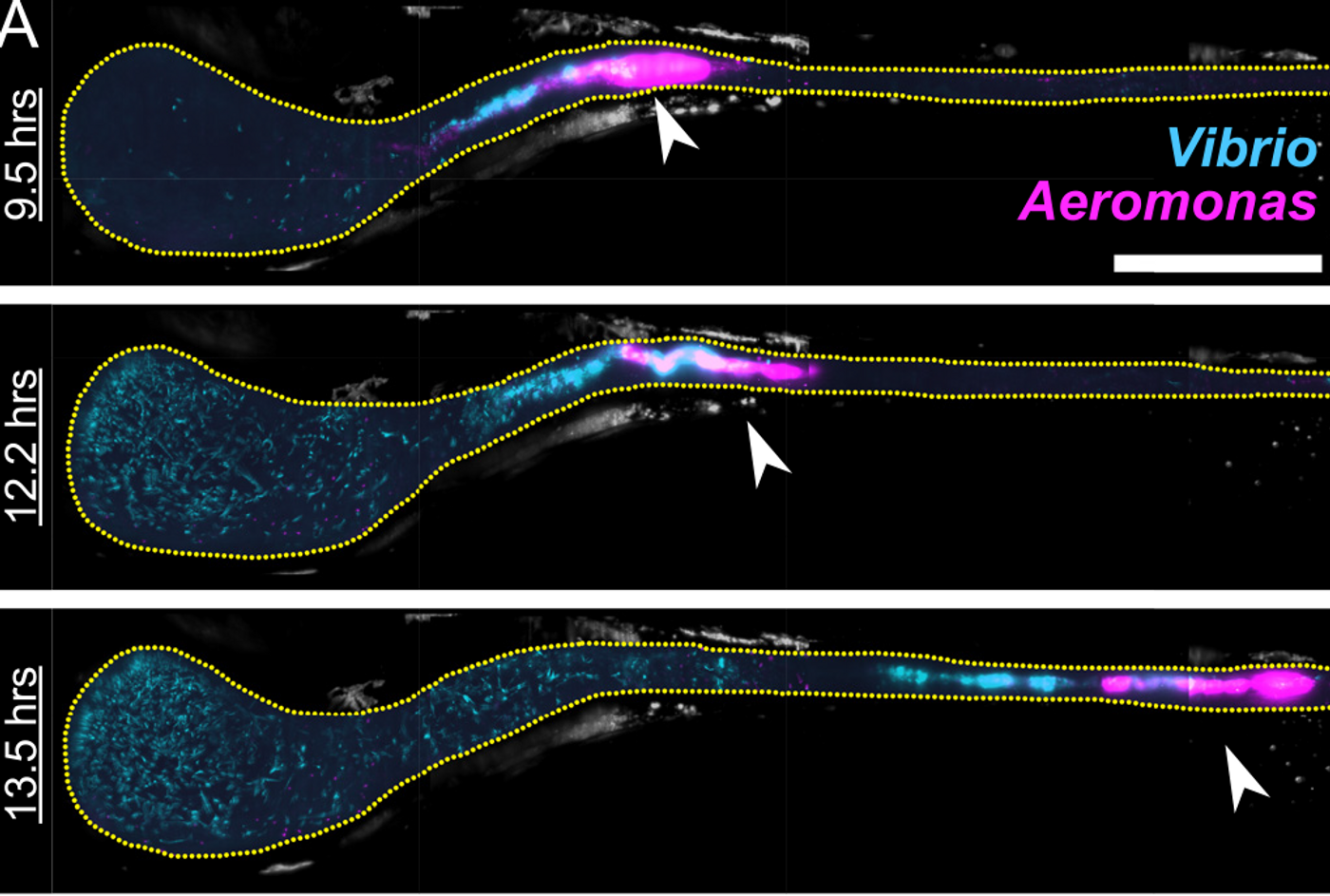
Publishing in open-access PLOS Biology, the scientists investigated two bacterial species native to zebrafish, Aeromonas veronii and Vibrio cholerae. The researchers showed that when zebrafish colonized by Aeromonas were subsequently exposed to Vibrio, populations of Aeromonas were reduced. The researchers wanted to know more about the mechanism driving that change.
The investigators took advantage of light-sheet fluorescence microscopy - an imaging technique that can quickly produce high-resolution 3-D images - and combined it with the benefits of live zebrafish. Images were captured every 20 minutes for 14 hours to precisely reconstruct the movement of bacteria in the guts of zebrafish.
Thus it was revealed, Aeromonas populations plummet, then rebound to normal growth rates whether exposed to Vibrio or not - although exposure to Vibrio did have an influence on the nature of those drops. The Aeromonas population collapses were tied instead to peristalsis -- the rhythmic contraction and relaxation of the muscles of the gut. The microbial species were reacting differently to the motions of the intestines. Vibrio "spread like a swarm of bees," Parthasarathy said, "and were unperturbed by peristalsis, while Aeromonas aggregated in particular locations that led to their expulsion."
The researchers used a model of Hirschsprung disease to learn even more. Hirschsprung is a birth defect in which nerve cells are absent from the ends of the bowels, and results in problems with bowel movements that are so severe, the condition is usually diagnosed in the days after birth. The video below explains more about the disorder.
In this work, the Hirschsprung model was so useful because it is an intestinal motility disorder, and would allow the researchers to see how the defects in that movement affected bacterial populations. They determined that there is no competition among bacteria in this system.
"The findings open a new window on problems occurring in patients with problematic peristalsis," said one co-author, Judith S. Eisen. "Our work provides new insights into these types of host-microbe interactions and may also prompt new thinking about therapeutic approaches for human disease."
Karen Guillemin, another co-author, explains, "about 30 percent of [Hirschsprung's] patients develop what is called Hirschsprung-associated enterocolitis, which is inflammation of the colon, and is likely to be a consequence of this altered microbiome. Our work provides new mechanistic insight in what could be causing the inflammation."
Want to know more about light-sheet fluorescence microscopy? Check out the ZEISS Webinar below.
Sources:
Mayo Clinic,
PLOS Biology,
AAAS/Eurekalert! via
PLOS