As research on the microbiome intensifies, new links between the microbial community the gastrointestinal tract and health continue to be established. It’s sometimes worth reiterating that links and correlation do not equal cause-and-effect, but it’s clear that the microbiome is critical to well being. Scientists at the Stowers Institute have added to this body of research with a
new study published in eLife. They show that the composition of the microbiome is related to the immune response of the host as well as the host’s ability to repair wounds.
Their work demonstrates that in the freshwater flatworm planaria, dramatic changes in its associated microbial community negatively impact tissue regeneration, which normally allows this microbe to re-grow parts. Such a microbiome change has a similar effect in humans as well, though that has not been replicated in model organisms like the fruit fly or zebrafish. The video above explains more about planaria regeneration.
The researchers pose this flatworm as a model for the study of the microbiome. Uncovering details about how immunity, the microbiome and tissue repair are related could be critical to therapies for chronic wounds.
"This is the first animal model to link pathological shifts in endogenous bacteria with the inhibition of regeneration," explained the senior author of the study Alejandro Sánchez Alvarado, Ph.D., of the Stowers Institute and the Howard Hughes Medical Institute. "We know that some kinds of bacteria are critical to our health, and that other kinds of bacteria can make it very difficult for us to recover from illness. Now we can study how the changing nature of the microbiome - and the way the immune system responds to those changes - impacts the natural execution of regenerative processes."
Researchers have been confounded by what seems to be dual roles for immunity in tissue regeneration. While the immune response promotes repair, it has also been shown to impede repair.
A problem that arose in Sánchez Alvarado’s laboratory presented an opportunity for study. An infection in the planaria population in the lab caused lesions to form near the eyes, then spread to the heads until they degenerated completely. Normally these worms would just grow a new head. But their regenerative powers were gone.
The steps the lab took to clear up the infection by circulating the water and sanitizing the growth media. But healthy worms removed from the system just got sick again, leading only to frustration for most lab members. But one saw a chance for analysis.
"I saw this as the perfect inducible model system. It was making lemonade out of lemons," explained Chris Arnold, Ph.D., a new postdoctoral research associate at the time. "We could take worms that were healthy, remove them from the tank system when desired, and place them in other conditions where they would then become ill. Amazingly, we found that when we withdrew the worms from the tank system and they developed problems, we could successfully treat their tissue degeneration with antibiotics. That suggested bacteria might be involved."
After identifying the bacteria residing with the flatworms, it was found that when the worms had lesions, there was a surge in one type of bacteria – Protobacteria. That led the researchers to wonder if it wasn’t the bacteria themselves affecting the worms’ ability to regenerate, but instead the immune system.
To get at that question, the investigators blocked genes that are part of the immune system in the worm. They found that when a gene called TAK1 was reduced, the worms were then able to recover from their infection. Looking at other genes that are part of the TAK1 pathway, they found others that had an effect on regeneration, but that only occurred when worms were infected.
"Our findings suggest that there is something special about regeneration during infection that's different than normal regeneration. There are genes that prompt degeneration in one case, and regeneration in another. It is topsy-turvy, completely different from what we would expect. We think this pathway might act to get rid of infected cells, clearing them out so the infection cannot spread to healthy tissue. Only when we block the pathway, can we allow regeneration to occur even in the presence of infection," said Sánchez Alvarado.
"Our healthcare system is struggling to deal with conditions of impaired wound healing. We know that bacteria are impediments to healing in patients, and that antibiotics aren't always effective, especially with the rise of antibiotic-resistant bacteria. By understanding the genes and pathways involved in the immune response, we may be able to interpret the signals that determine whether an organism decides it is beyond repair or tries to regenerate. Perhaps then could we develop more effective therapies," concluded Arnold.
Sources:
eLife,
AAAS/Eurekalert! via
Stowers Institute
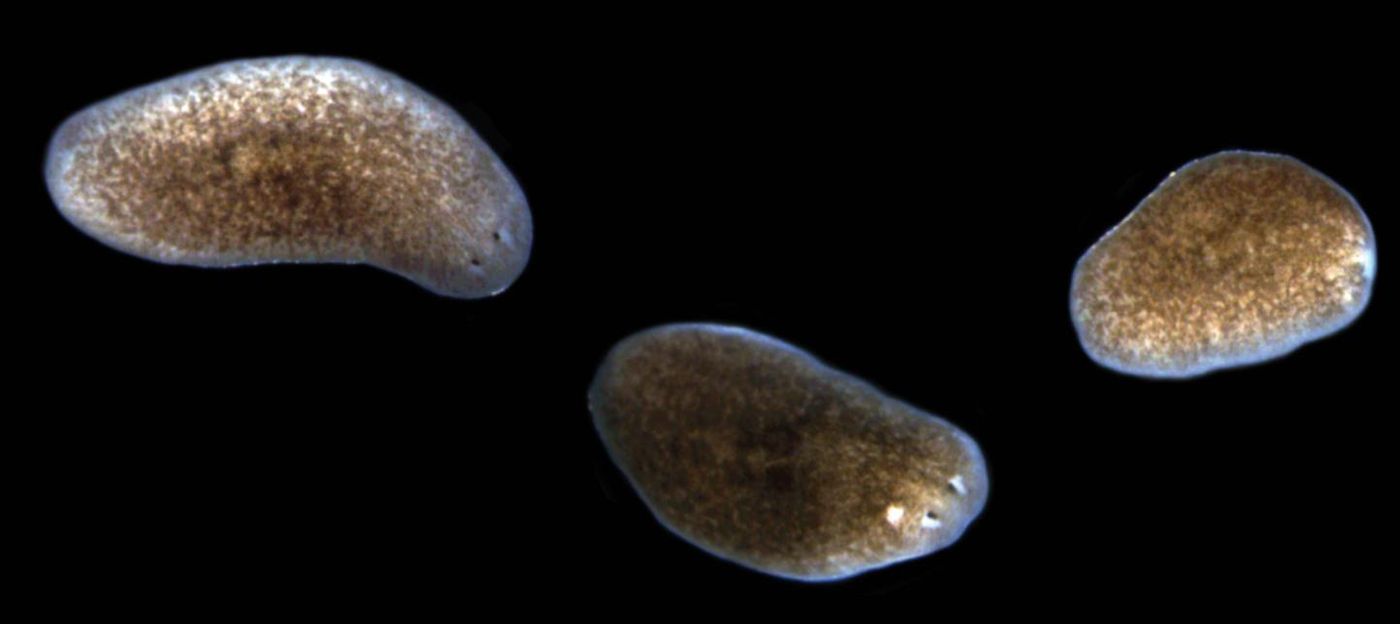
![Representative images and median pathological stage of worms following [gene knockdown] with each combination of 6 activators and 3 inhibitors 12 days post Pseudomonas infection / Credit: eLife, Arnold et al](https://d3bkbkx82g74b8.cloudfront.net/eyJidWNrZXQiOiJsYWJyb290cy1hc3NldHMiLCJrZXkiOiJfcHVibGljXC9fZmlsZXNcL3N5c3RlbVwvY2tcL3RyZW5kaW5nXC9wbGFuYXJpYTJfODIzMWJiYWI1M2U2ZDBiOTdiNDk2NWIxYmU5NWZiOWMuanBnIiwiZWRpdHMiOnsicmVzaXplIjp7IndpZHRoIjoxNDAwLCJmaXQiOiJjb3ZlciJ9fX0=)