Aiming to improve the treatment for cystic fibrosis (CF), researchers at Caltech have created new tool for visualizing infections in the lungs. The buildup of thick mucous called sputum in the lungs is one of the hallmarks of CF. The new technique, called MiPACT, makes the septum transparent as well as colorizes different cell types so a three dimensional “biogeographical map” of a lung infection can be built for a CF patient. The map allowed the investigators to see that the majority of bacteria in these lung infections are in a state of slow growth and because of that physiology, cannot be treated with most antibiotics.
The MiPACT technology was described in the video above and in
a report published in mBio by the research teams of Dianne Newman, a Professor of Biology and Geobiology and HHMI Investigator, and Viviana Gradinaru, an Assistant Professor of Biology and Biological Engineering. While it was tested for use in the treatment of CF, it has the potential for broad applications.
"The National Microbiome Initiative, launched earlier this year by the White House Office of Science and Technology Policy, specifically called for new methodologies to study microbial communities across diverse environments," adds Gradinaru. "The MiPACT technology is extremely versatile and can be applied to many topics in parasitology and microbiology: wound infection, biofilm formation, microbe distribution within a niche, or host-microbiome interactions such as in the human gut."
This work builds upon a technique created in 2014 by Gradinaru’s lab called PACT or Passive CLARITY that makes organs and firm tissues of the body transparent. Newman, studies the nature of microbial survival in the context of infections, and was inspired by Gradinaru's work. "It hit me that maybe this could be applied to the things we work on in my lab—namely, cystic fibrosis sputum," said Newman. "We had been studying an important CF pathogen in my lab for many years, but found ourselves increasingly motivated to determine whether our laboratory findings were relevant in actual infections. We literally needed a new way of seeing."
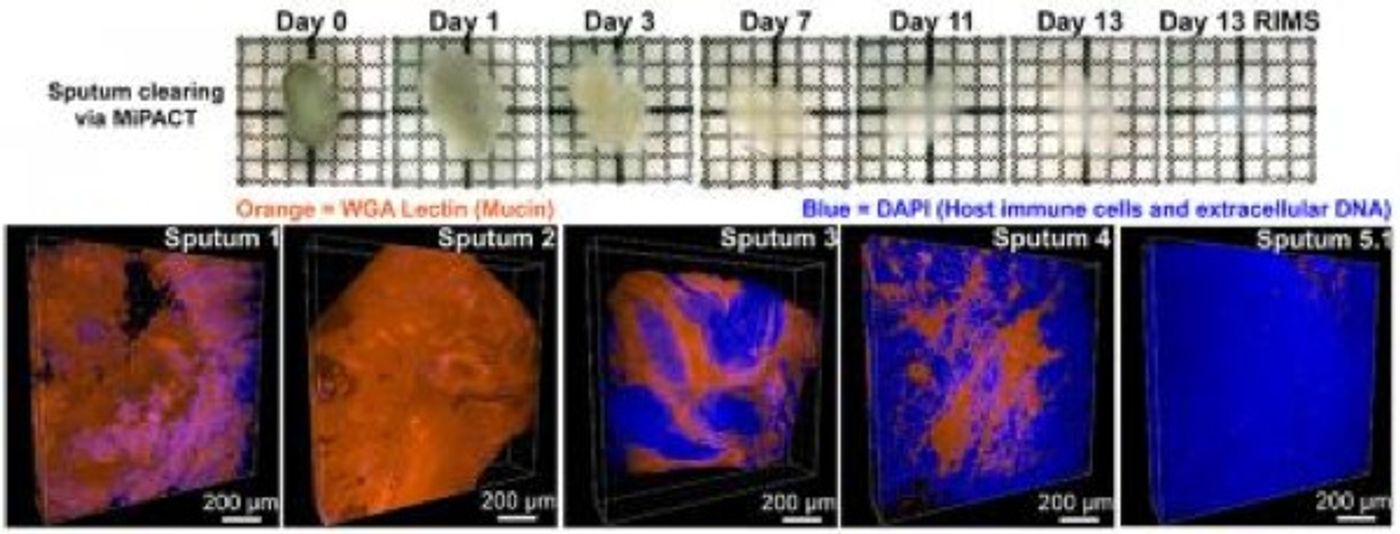
Lipids create opacity in things like bones and phlegm because they scatter light. The PACT method removes the lipids, and biological samples are thus made transparent. However, sputum doesn’t have the same structure as most tissue because it’s not a grouping of cells. The PACT technique needed a tweak.
"PACT was developed for tissues that already have some 3-D structure and consistency, such as the brain or heart," Gradinaru explained. "Sputum is challenging because it does not have this structure. With MiPACT, we employed a thick hydrogel that we can infuse into a sample of sputum, locking bacteria, cells, and other components in place for subsequent detection."
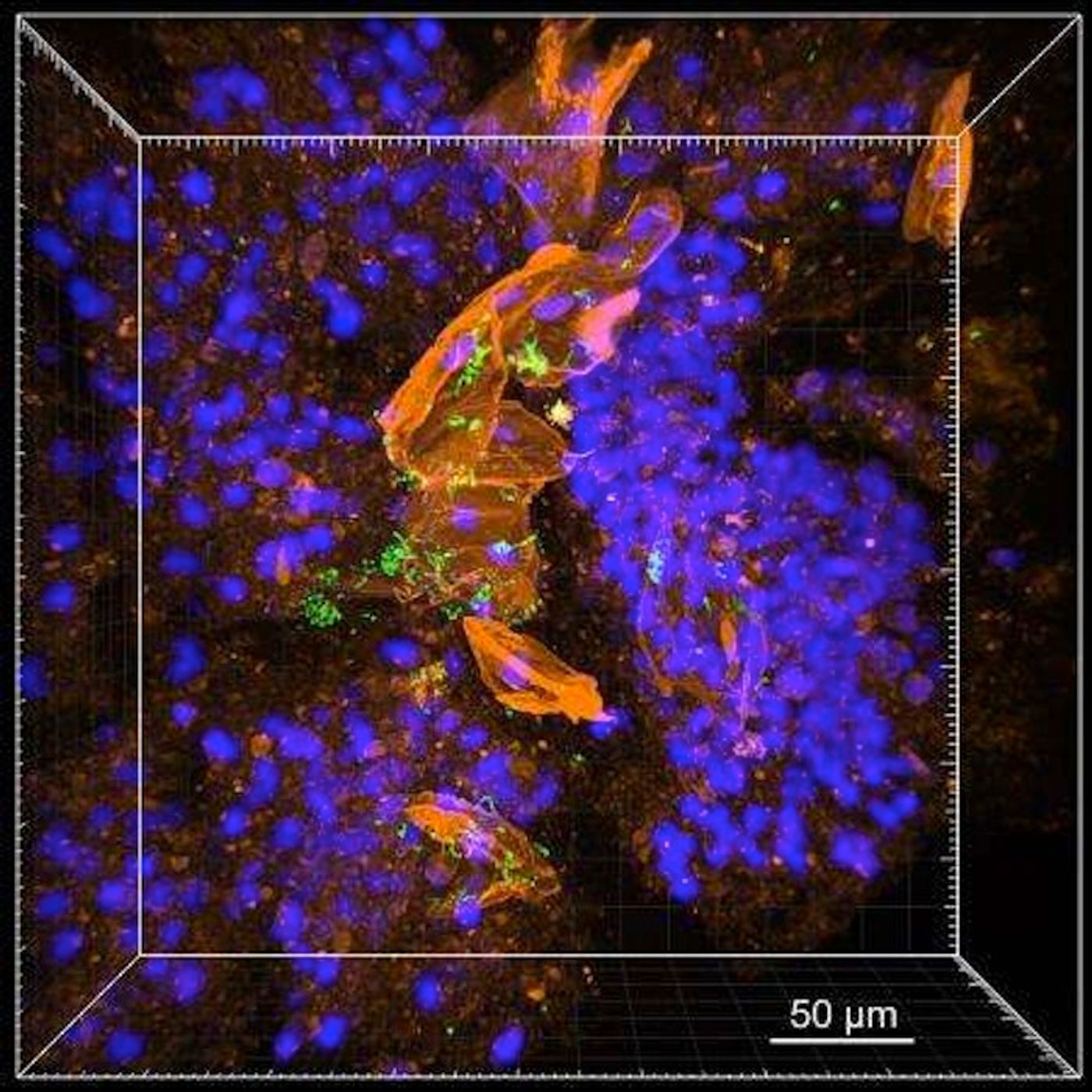
Now, various components are kept undisturbed; the lipids can subsequently be removed to leave the sample totally transparent and completely intact. Finally, fluorescent probes are applied to the sample, and bacterial cells can be easily seen. That could be a great help to those suffering from CF.
"Cystic fibrosis research is often focused on Pseudomonas aeruginosa—which is the dominant pathogen in the lung near the end of a patient's lifespan," Newman commented. "However, there are many important pathogens in the early stages of the disease—for example, children with cystic fibrosis are more likely to have Staphylococcus aureus. Each of these pathogens have distinct aggregation patterns. It's important to determine how these different bacteria behave in order to design experiments that come as close as possible to capturing the actual conditions of chronic infection."
MiPACT can be used with large amounts as well; researchers studying at sputum can thus use a sample of the environment in the lungs that is accurately represented. The scientists stress that the tool can be applied to the investigation of other microbial environments such as the human gut.
"We hope that MiPACT will enable the study of microbe-host interactions in diverse contexts. One meaningful area of potential application would be to better understand the structure and function of beneficial microbial communities, such as those comprising the human microbiome," concluded Newman.
Sources:
Caltech,
mBio