Silver to the rescue!
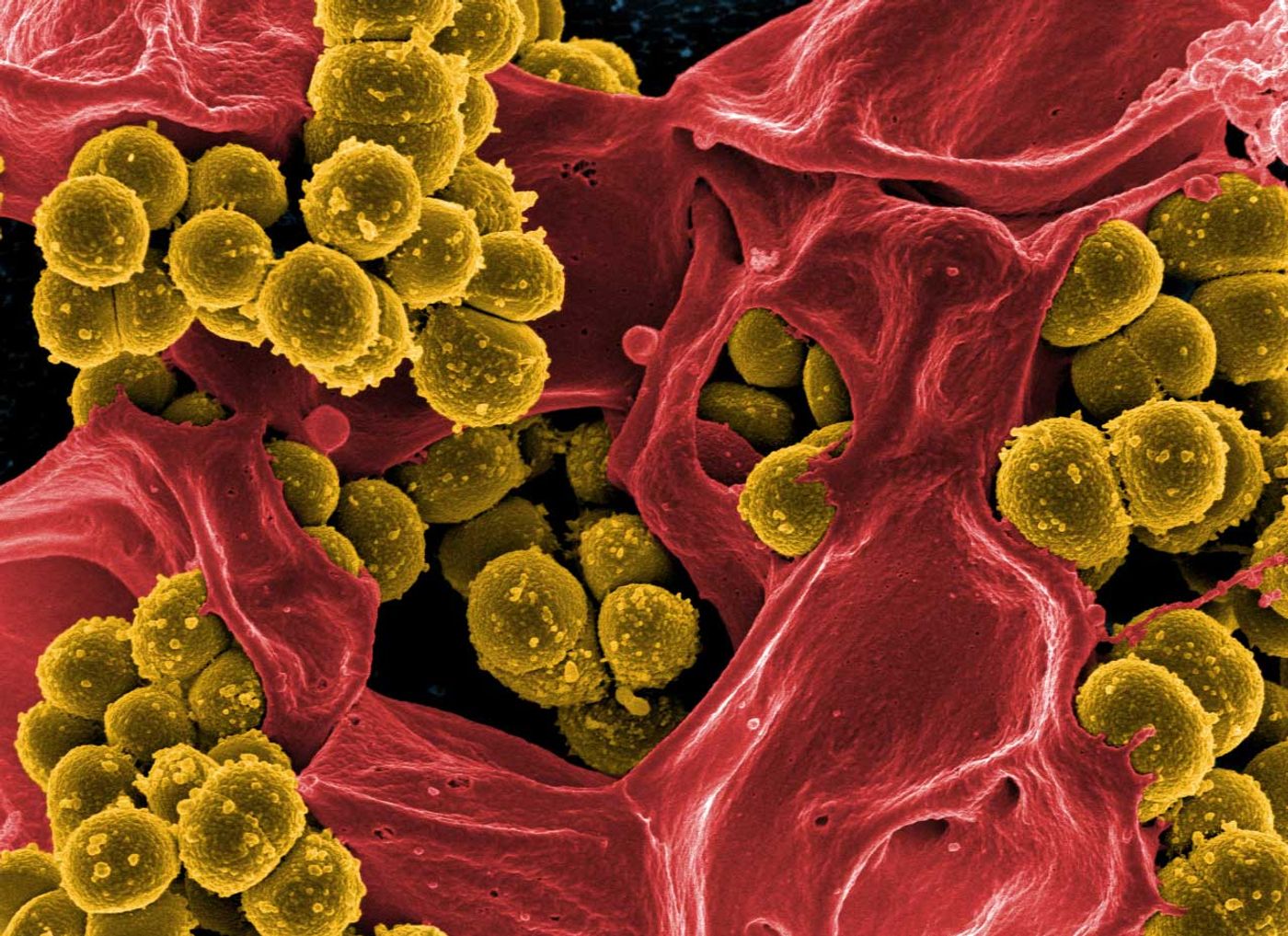
Osteomyelitis is a serious bone infection, often caused by the bacterium Staphylococcus aureus. These infections are difficult to treat because S. aureus forms tough biofilms and is often resistant to antibiotics - take MRSA, for example.
Osteomyelitis can occur when bacteria from the blood or nearby tissue infiltrate bone. Alternately, implanted medical devices, such as artificial joints, can be contaminated with bacteria. Most cases of osteomyelitis require a surgeon to remove portions of the infected bone, and patients require intravenous antibiotics for up to six weeks.
A collaborative group of investigators from the University of North Carolina at Chapel Hill, North Carolina State University, Silpakorn University in Thailand, and the University of Missouri, Columbia, previously designed a biocompatible scaffold that contains silver ions - designed to kill bacteria. The scaffold is made up of polylactic acid (PLA) nanofibers and coated with a proprietary polymer that contains silver nitrate.
Silver, among other metals, has been used to kill bacteria or prevent their growth for centuries - this phenomenon is called the “oligodynamic effect”. The precise mechanism behind silver’s antibacterial activity remains unclear, however. Silver may bind to and inactivate proteins within bacterial cells, or it may damage the cell wall of bacteria. Either way, it is considered relatively non-toxic to mammalian cells.
The researchers hope the scaffolding could be placed on infected bone to help fight infection and promote healing. The scaffold could also be used during orthopedic surgery to prevent infection. The first step however, was to see if the silver-seeded scaffold could actually kill bacteria.
The researchers placed either a control PLA scaffold (without silver) or a PLA scaffold with silver onto a lawn of MRSA. Only the PLA scaffold containing silver killed the surrounding bacteria, producing a clear zone of inhibition around the scaffold.
Next, they added human adipose-derived stem cells (hASCs) to the mix. These cells were added to the scaffold and induced to differentiate into bone cells. When MRSA was cultured with the PLA scaffold containing hASCs, but no silver, the bacteria proliferated rapidly over 2 days, and continued to proliferate steadily for 2 weeks. If silver was added to the mixture of PLA, hASCs, and MRSA, the bacteria proliferated much more slowly over the first 2 days, but proliferation picked up at day 5 of 13.
Although the results of their second experiment were not as impressive as their first, these data demonstrate that a silver-seeded scaffold could help resolve, or at least slow, cases of osteomyelitis. According to journal editor Peter C. Johnson, “hybrid therapeutic approaches such as this combination of a regenerative and anti-infective platform are transforming our attack on complex musculoskeletal diseases.”
Sources: Tissue Engineering, MicrobeWiki, EurekAlert, Mayo Clinic, Wikipedia