The power of microbial fuel cells
A microbial fuel cell is exactly what it sounds like, a fuel cell powered by microbes.
I have to admit, I’d never heard of a microbial fuel cell until recently (or maybe I had, but it just didn’t register).
The first microbial fuel cell (MFC) was rigged up by M.C. Potter way back in 1911 - this MFC used E. coli to generate electricity. Unfortunately, the idea didn’t gain much traction until the 1970s. At that time, Robin M. Allen and H. Peter Bennetto put their heads together to figure out how MFCs actually work.
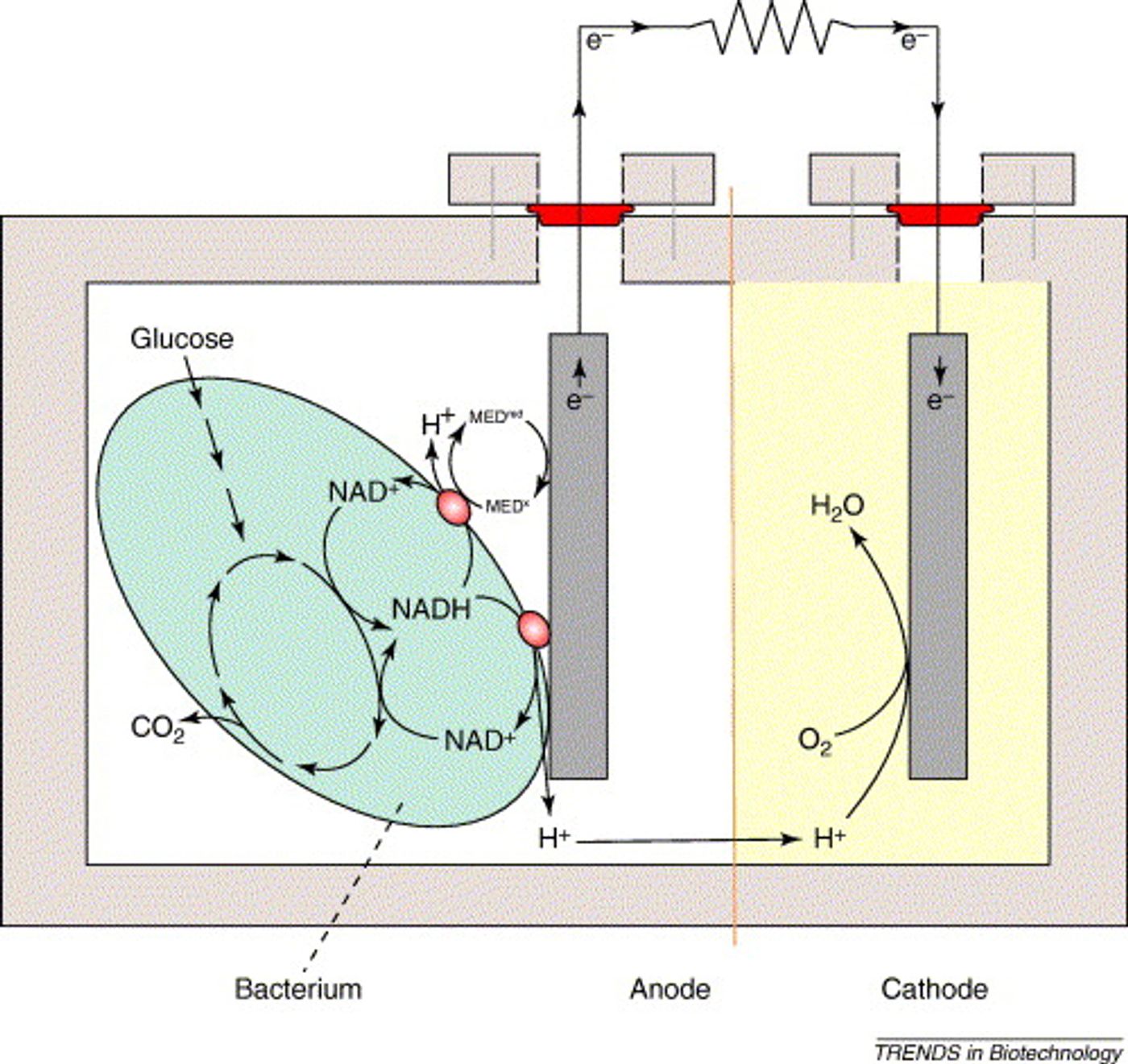
Very simply, an MFC uses bacteria to convert chemical energy to electrical energy. In many MFCs, that chemical energy is glucose. When bacteria metabolize glucose under aerobic conditions, they produce carbon dioxide and water. However, under anaerobic conditions, they produce carbon dioxide, protons, and electrons. The electrons are the key to an MFC.
The generic MFC has one chamber containing bacteria and an anode. The anode is connected by a wire to the cathode in a neighboring chamber. The electrons pass from the bacterial electron transport chain to the anode. (There are a number of ways to transfer electrons from the bacteria to the anode, but I won’t bore you with the details.)
The cathode chamber can simply contain air (assuming your air contains oxygen). Here, the oxygen acts as the electron acceptor - electrons leave the cathode and combine with oxygen to produce water. (Yes, this also requires hydrogen ions. These are also produced by the bacteria and can cross a membrane or salt bridge that separates the anode and cathode chambers.)
The basic MFC I’ve just described is called a “mediator-free” MFC. This means that the electrons are passed directly from the bacteria to the anode - they don’t need a mediator. (Various chemicals can be added to the anode chamber to act as mediators.)
Mediator-free MFCs are way more cool that mediated MFCs. They use so-called electrochemically active bacteria that transfer electrons directly to the anode. Many of these bacteria produce pili or “nanowires” to shuttle electrons from their outer membrane to the anode. (Yes, this is as cool as it sounds.)
MFCs can run on a number of chemical energy sources, but my favorite has to be soil. In its simplest form, a soil-based MFC uses soil as the anode. The soil provides the bacteria, the chemical energy source, and acts as the medium for proton exchange (how convenient!). Simply place your anode down in the soil, and leave the cathode at the surface, exposed to the air. You can even build your own soil-based MFC at home!
Okay, are these MFCs just a fun party gag? Or could they really be used to generate electricity on a large(ish) scale?
It turns out that MFCs are quite widely applied to wastewater treatment. These systems generate energy in the process of decontaminating wastewater. How about the oil and gas industry’s wastewater? That stuff contains lots of hydrocarbons and lots of salt. The hydrocarbons can be metabolized by bacteria, producing electrons that power desalination!
Thanks, bacteria!
Sources: Environmental Science and Technology, New Atlas, Pennsylvania State University, Wikipedia