Better Know a Microbe: Geobacter
Geobacter is a remarkable genus of bacteria. They were the first organisms found to be able to oxidize organic compounds and metals into carbon dioxide. In other words, these bacteria use metals to “breathe” in the same way that we use oxygen!
The first Geobacter specimen was identified in 1987 by Derek Lovley. The bacteria were isolated from sediment in Washington D.C.’s Potomac River. Since then, we humans have been working to use Geobacter for our own selfish benefit (because that’s what we do best).
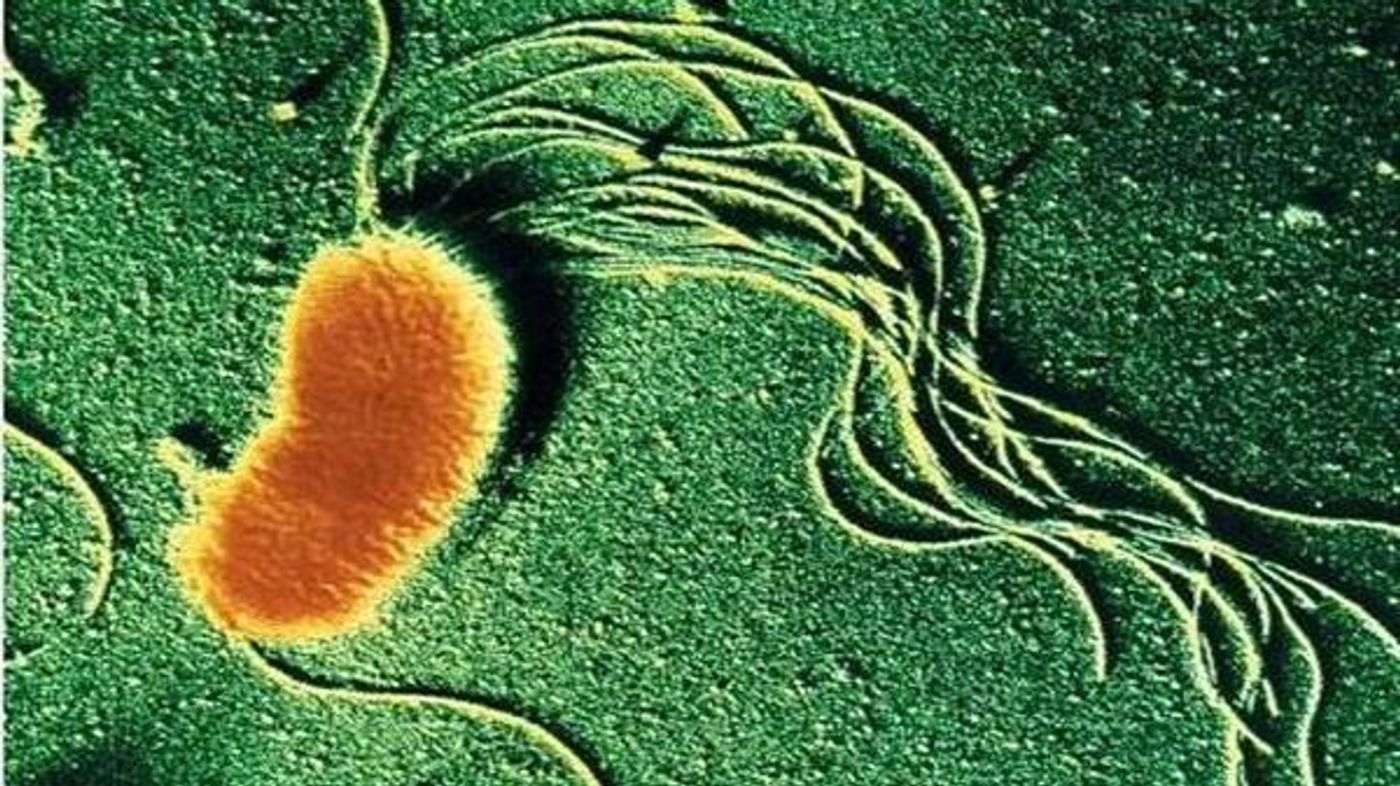
So, here are some fun facts about Geobacter and an introduction to some of the ways we use Geobacter. I’ll start you off with the basics. Geobacter species are anaerobic, rod-shaped, Gram-negative bacteria that produce flagella and pili.
The first fun fact is that much of the magnetite that is associated with ancient iron deposits was probably produced by Geobacter. That’s because Geobacter reduces Fe(III) to magnetite (Fe3O4).
In fact, Geobacter often uses Fe(III) as an electron acceptor, but what happens when the Fe(III) runs out? No problem, Geobacter can form a multispecies aggregate to help generate energy. When the Fe(II) runs out, the Fe(III)-reducing bacteria form syntrophic associations with bacteria that can accept electrons that would normally be transferred to Fe(III). This concept was also demonstrated in an experiment with ethanol and sodium fumarate.
In this case, two species of Geobacter cooperated to metabolize ethanol and sodium fumarate. One of the species, G. metallireducens, metabolized the ethanol to produce an excess of electrons. These electrons were sent over to G. sulfurreducens with nanowires that grew between and linked the bacterial cells. These electrons allowed G. sulfurreducens to break down the sodium fumarate.
Geobacter can also oxidize petroleum products, making these bacteria especially useful for cleaning up water and soil that is polluted with oil. Since Geobacter also oxidizes metals, these little guys are also great at cleaning up sites that are contaminated with radioactive metals.
Another fun fact - nanowires! Some Geobacter species make nanowires composed of proteins with metal-like conductivity. As the name suggests, nanowires are only 3 to 5 nanometers wide - that’s 10,000 times smaller than the width of a human hair! The nanowires can also grow to be some 20 nanometers long - more than 10 times as long as the bacterial cell.
Because of these nanowires, G. metallireducens can be attached to an electrode to produce an electric current. The nanowires conduct electricity and could potentially be used as a sustainable electronic material. Microbial nanowires could be easily mass-produced at low cost. This is compared to manmade conductive materials that require expensive and toxic reagents. According to Geobacter guru Derek Lovley, “microbial nanowires therefore offer an unprecedented potential for developing novel materials, electronic devices and sensors for diverse applications with a new environmentally friendly technology. This is an important advance in microbial nanowire technology.”
The nanowires made by G. sulfurreducens may actually play double-duty - there’s evidence that in addition to conducting electric current, they have a structural role in biofilm formation. Researchers grew the bacteria on a coverslip and gave them fumarate as an energy source - the bacteria don’t have to use nanowires to get energy from fumarate. Despite this, bacteria that couldn’t produce pili didn’t grow as well. The group found that the pili help the bacteria to agglutinate, which helps them to form a biofilm. They measured the optical density of cultures, and found that cells that lacked the genes for producing pili did not agglutinate. If they complemented this mutant by expressing the pilin genes from a plasmid, the cells agglutinated.
Sources: Geobacter Project, Design News, Small Things Considered, Science, Journal of Bacteriology