Better Know a Microbe: Yersinia pestis
If you’re not familiar with Yersinia pestis, that’s okay. However, I’m sure you’re familiar with the plague. Does the Black Death ring a bell?
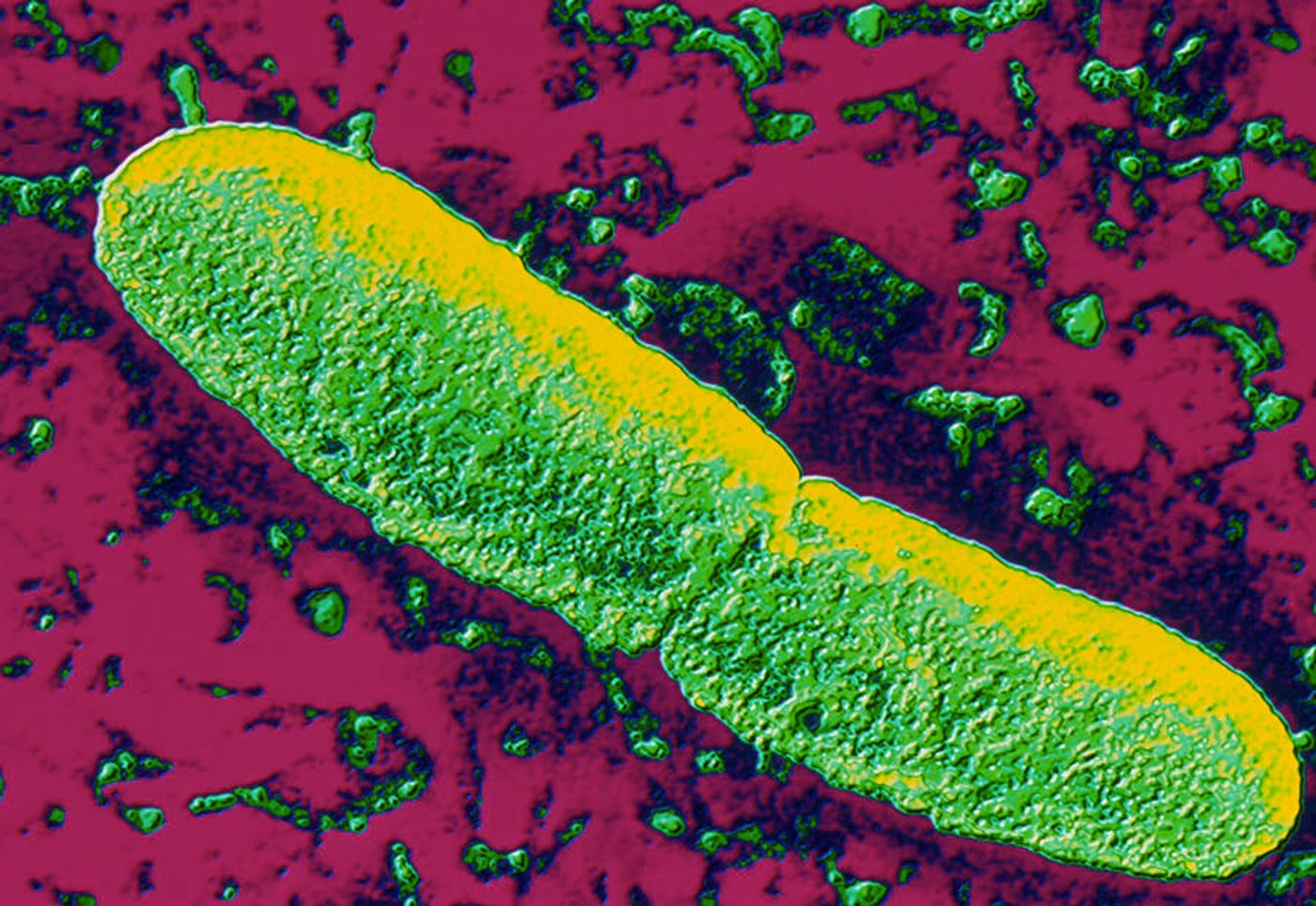
The plague actually comes in three flavors - bubonic, pneumonic, and septicemic. All three are caused by the bacterium Yersinia pestis, a rod-shaped coccobacillus. This pesky pathogen was discovered by Alexandre Yersin who was working at the Pasteur Institute. At the time, a plague outbreak was occurring in Hong Kong, and the Japanese bacteriologist Kitasato Shibasaburo was also working to identify the cause of plague.
The most notable outbreaks of plague are the 6th century Plague of Justinian, the 14th century Black Death, and the 19th century’s Third Pandemic. The Plague of Justinian occurred from roughly 541-542 AD and killed nearly one-third of the population of the Byzantine Empire. This plague was probably caused by a now-extinct strain of Y. pestis. The Black Death gets all of the press, however, and rightly so. This epidemic was most severe in the 1340s, but continued to plague (see what I did there?) Europe for some 400 years. The Third Pandemic occurred in Yunnan province in China in 1855. This round of disease reportedly spread to all inhabited continents, killing some 12 million people in China and India.
Do you think plague is a disease of the past? Think again - the World Health Organization still reports thousands of cases each year. Of course, this pales in comparison to previous epidemics, but we’d do well to remember that this pathogen is still very present. Today, most cases of the plague occur in sub-Saharan Africa and Madagascar. The disease was introduced to the US in 1900 when rat-infested ships sailed in from Asia; the last outbreak in the US occurred in Los Angeles from 1924 to 1925.
Bubonic plague causes fever, muscle ache, gangrene, and the characteristic buboes - severely swollen lymph nodes. People can die from this condition in less than 2 weeks. Septicemic plague occurs when bacteria enter the bloodstream. In these cases, people experience hepatosplenomegaly, delirium, and general malaise. Unfortunately, people can die before these symptoms ever appear. As for pneumonic plague, this lung infection causes coughing, chest pain, hypotension, and hemoptysis (coughing up blood). This condition is always fatal if not treated.
If diagnosed early on, plague can be treated with antibiotics such as streptomycin, chloramphenicol, tetracycline, and fluoroquinolones. However, a handful of resistant strains are circulating. There used to be an inactivated vaccine for plague available in the US, but it was removed from the market - it was not very effective and it often caused severe inflammation.
How do we humans end up with the plague? It all starts when Y. pestis infects fleas. The fleas pick up the bacteria by feeding on an infected host. Then, these fleas hang out on rodents, usually the brown rat (although squirrels, chipmunks, and prairie dogs are also carriers). One thing leads to another, and rodents and humans end up living in close proximity (these conditions are exacerbated by substandard living conditions). From there, the infected fleas bite us humans, and us humans get the plague. Your best defense against the plague is to avoid rodents, especially in areas where rodent plague is endemic - the Southwest US, for example.
While Y. pestis is definitely bad for humans, it’s not all that nice to fleas either. The bacteria produce hemin storage proteins when they are in their insect host to help them move back to a mammalian host. The proteins induce biofilm formation in the flea’s proventriculus, a valve that connects the esophagus and midgut. When the biofilm grows large enough that the flea can’t feed, they regurgitate it back into the bloodstream of whatever mammal they’re feeding on. See, don’t you feel kind of sorry for the fleas?
Last but not least, because Y. pestis is able survive inside of human macrophages, and human macrophages are pretty similar to amoebae, researchers at Washington State University set out to see if Y. pestis could survive inside of amoebae.
They cocultured different strains of Y. pestis with amoebae, killed any extracellular bacteria, lysed the amoebae, and finally cultured this lysate. They found that some strains of Y. pestis were able to survive inside of the amoebae for over 5 days! The researchers knew that the protein PhoP helps Y. pestis survive inside of macrophages, so they tested whether the same protein helped the bacteria survive inside of an amoeba. Sure enough, a PhoP mutant could not survive inside of the amoebae.
Sources: CDC, Applied and Environmental Microbiology, Science Daily, Wikipedia