Understanding How Tuberculosis Takes Hold
New work by researchers at the University of Cambridge has provided insight into how tuberculosis infections get a foothold and begin to grow. Mycobacterium tuberculosis is the bacterium that causes the illness, which is a leading cause of death worldwide but impacts fewer than 200,000 people annually in the United States. According to the World Health Organization, while 95 percent of tuberculosis deaths happen in low-income countries, infections that are resistant to treatment are on the rise. Learning more about the disease is vital if we are to control outbreaks.
The new research, published in the journal Cell, may help to prevent tuberculosis infections by helping scientists develop better treatments or vaccines. The video is the abstract for this work, by the authors.
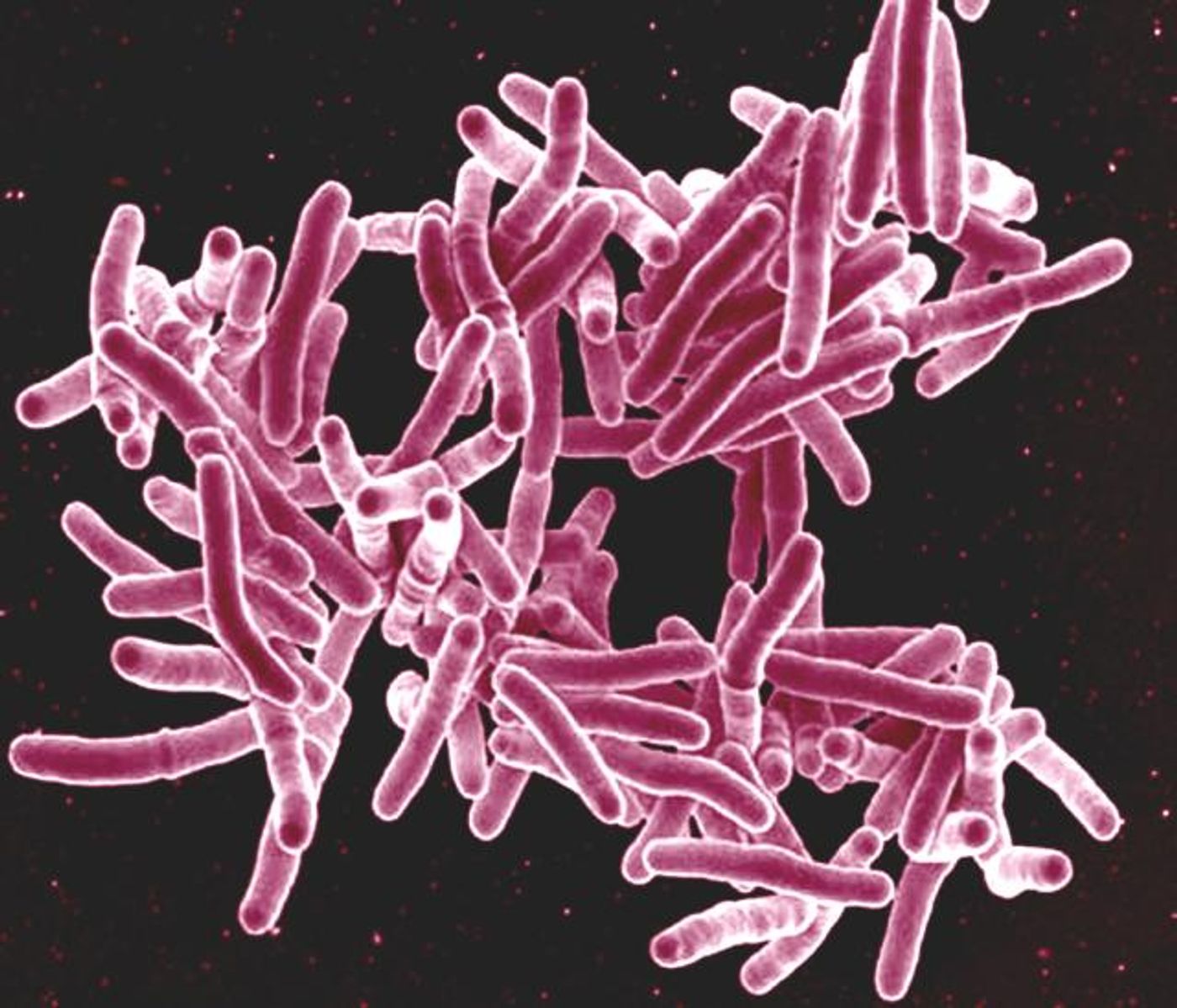
The Mycobacterium tuberculosis (Mtb) bacterium enters the lung, where it encounters macrophages that reside there. These macrophages are important immune cells are part of our first line of defense against invaders. They work to remove particles and microbes from the air we breathe.
A zebrafish model demonstrated that the pathogenic Mtb gets taken up by these lung-resident macrophages, and then destroyed. That suggested that for an infection to be established, the bacterium has to escape the resident macrophages, and into monocytes. The monocytes will allow growth of the bacterium, which is then able to get an infection going.
This was also the case for another pathogen that the researchers analyzed, Mycobacterium leprae, the bacterium that causes leprosy.
"These 'Pac-Man'-like immune cells swallow the leprosy bacteria, but are not always able to destroy them," explained senior author of the new report, Lalita Ramakrishnan, a Professor at the Department of Medicine at the University of Cambridge. Ramakrishnan’s lab is part of the Medical Research Council's Laboratory of Molecular Biology. In the case of leprosy, the bacterium impacts nerves. "Instead [of destroying the bacteria], the macrophages -- which should be moving up and down the nerve fiber repairing damage -- slow down and settle in place, destroying the myelin sheath."
In tuberculosis, Mtb is helped along by a molecule called mycobacterial membrane phenolic glycolipid (PGL), which is crucial to enabling the transition from macrophage to monocyte. PGL activates a pathway which eventually leads to the recruitment of monocytes for the establishment of an infection. The investigators believe that PGL now presents a therapeutic target.
Sources: Cell, Science Daily, World Health Organization