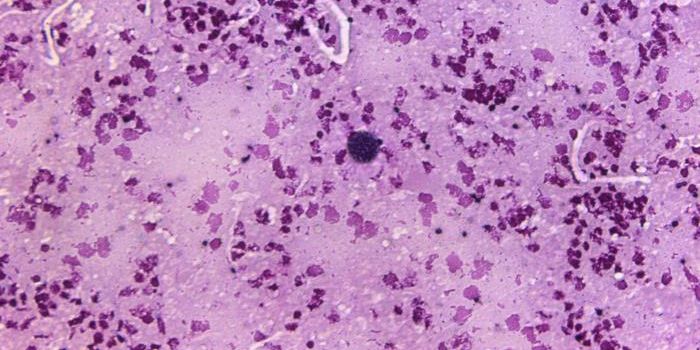
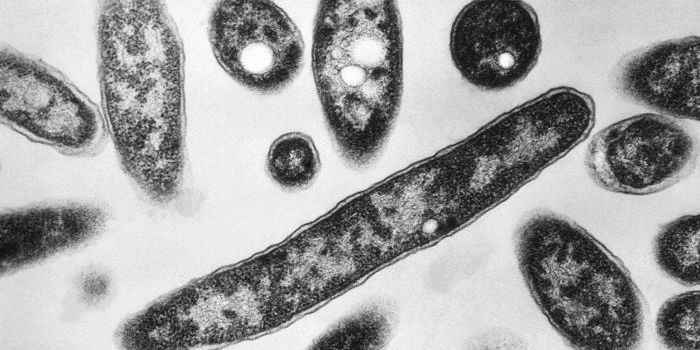
Bacteria form multicellular symplasmata
Bacterial multicellularity sounds like an oxymoron, right? Actually, bacteria in the genus Pantoea (family Enterobacteriaceae) assemble into multicellular structures called symplasmata. These structures can contain hundreds of cells and are surrounded by a polysaccharide capsule - they look sort of like a giant cell filled with lots of little cells!
Image: Of Bacteria and Men, Tecon and Leveau
Pantoea live in most all environments and colonize various animal, plant, and insect hosts. As individual, free-living cells, they look a lot like E. coli - they’re rod-shaped, Gram-negative, and can swim around using flagella. Interestingly, each symplasmatum starts with just a single bacterium, meaning that all of the cells in the structure are clones. These structures can also form independently of a solid surface, unlike a biofilm.
What’s the benefit of living the multicellular life? Safety in numbers? I’m imagining a crowded apartment building and having trouble seeing the pros (I mean who really wants a roommate?). Some reports suggest that these unique structures protect bacteria from environmental stresses like heavy metals, UV, and acids.
For Pantoea that colonize plants, symplasmata form on leaves and roots and even within the plant’s tissues. For P. eucalypti (a plant colonizer), a typical symplasmatum can contain anywhere from a few cells to hundreds - Tecon and Leveau penned a comprehensive review of P. eucalypti (Pe) symplasmata. Pe cells inside the symplasmata are often irregularly shaped, and they form cell-to-cell contacts with neighboring cells - the cells are actually more tightly packed in a symplasmatum than in a biofilm.
What does it take to make a symplasmatum? Tecon and Leveau reported that cells growing in liquid minimal medium with glucose readily formed symplasmata at 30oC and pH 7.2. On the other hand, Pe did not form symplasmata if they were grown in rich medium or at warmer temperatures. In fact, cells inside the symplasmatum can perceive and react to external signals. When they get a signal to leave the symplasmatum, they do so in rather explosive bursts! If you can access the Tecon and Leveau paper, take a look at the video of cells exiting their symplasmatum to replicate. (I couldn’t stop watching!)
In the same study, Tecon and Leveau used transposon mutagenesis to identify Pe genes that are required to form symplasmata. Transposon insertions in the gene lrhA totally abolished symplasmatum formation; LrhA is a regulatory protein that suppresses flagellar motility. Similarly, insertions in cspD or rfbX3 reduced the production of symplasmata.
This group also investigated the transcriptional profile of cells in symplasmata. Interestingly, genes for the uptake and degradation of specific carbon sources were upregulated - the same carbon sources that make up the symplasmata capsule.
Jiang et al. investigated the contribution of quorum sensing to symplasmata formation in P. agglomerans (strain YS19), endosymbionts of rice. The most common quorum sensing molecules for bacteria are acyl-homoserine lactones (AHLs)
The researchers first verified that P. agglomerans (Pa) indeed produces an AHL, and they went on to identify it as N-3-oxooctanoyl-L-homoserine lactone (OOHL). The big question, of course, was does OOHL promote symplasmatum formation? Indeed, adding OOHL to the growth medium significantly increased the size of symplasmata and the rate at which they formed. Because OOHL also increased Pa’s swimming and swarming behavior, the researchers reasoned that motility helps promote symplasmatum formation.
Oddly enough, when Pa colonizes rice plant tissues, it’s almost exclusively in the form of symplasmata. Thus, the researchers reasoned that OOHL may be especially importantly for Pa to colonize rice. Sure enough, OOHL increased Pa colonization by almost 67%. On top of that, the combination of OOHL and Pa increased the growth of the rice plants. Oxymoron or not, bacterial multicellularity has its benefits!
Sources: Nature, Journal of Basic Microbiology, Of Bacteria and Men