Towards the Creation of Living Machines
A special bacterium can function as a kind of miniature natural power plant, researchers say. A species of microbes called Shewanella oneidensis have a unique metabolism in which they interact with rocks in a way that is comparable to how we breathe. Now, Moh El-Naggar, an associate professor of physics, biological sciences, and chemistry at USC Dornsife and a team from USC and Caltech have learned how these bacteria transfer electrons across their membrane with a kind of nanowire.
This work, reported in the Proceedings of the National Academy of Sciences, may help advance a new kind of technology that uses microbes to generate energy. Bacteria that feed on organic material or other stuff like rust can create electricity, as explained in the video. Another kind of microbe being developed at USC can feed on waste, oxidizing organic substances in wastewater to generate a bit of electricity in the process.
"Microbes are highly evolved machines," El-Naggar said. "And what we have here is a class that is really good at converting energy and interacting with the abiotic world."
These organisms also provide a window into how and what life might survive in places that lack oxygen, like in outer space or deep in the ocean.
They live by moving electrons out of the cell, said El-Naggar. "If one were to shut down the ability to transfer the electron out of their system, they would not be able to make energy. The bacteria would basically suffocate," he explained.
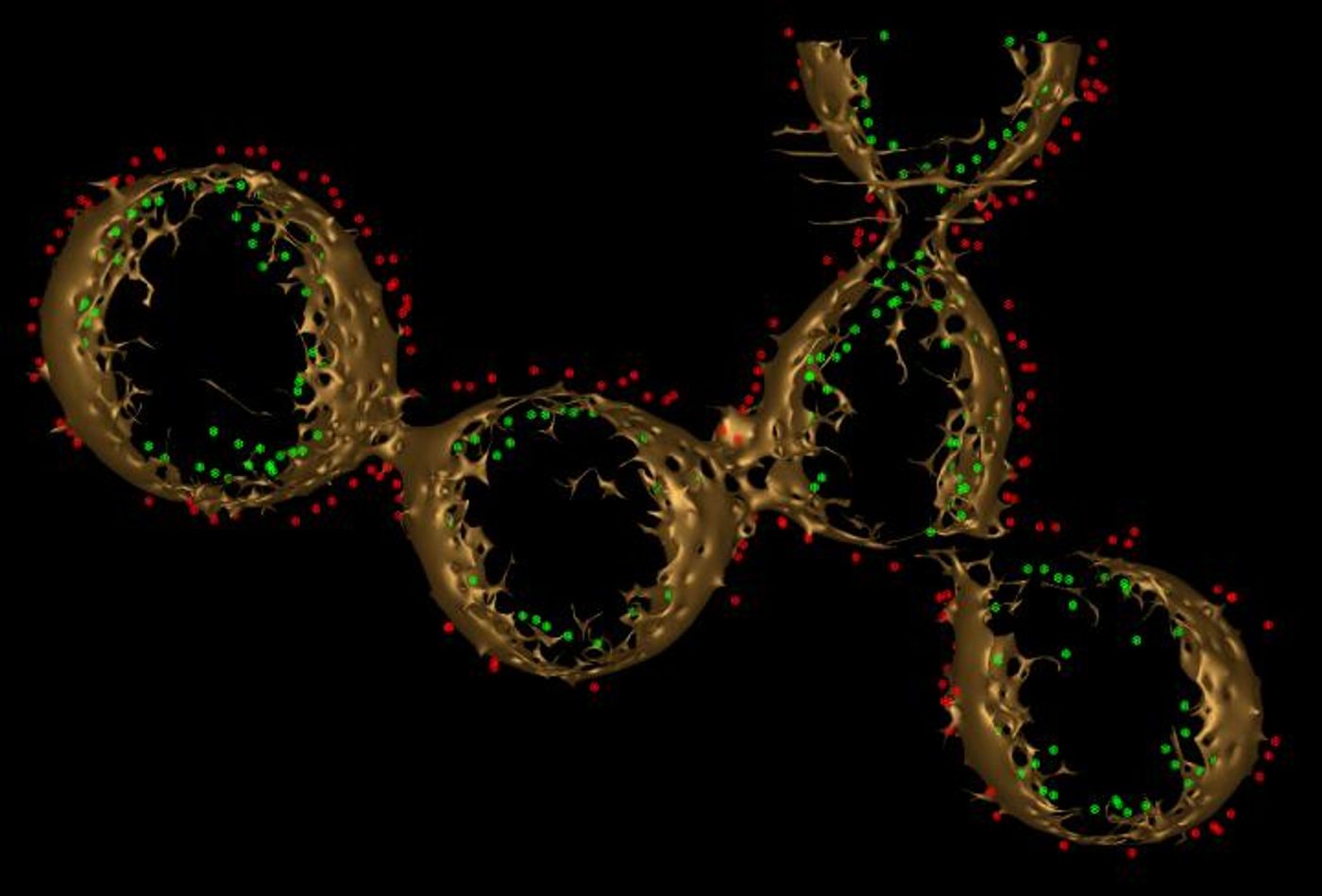
These bacterial cells also have protruding filaments, which were thought to be a microbial feature called pili. In 2013, researcher Sahand Pirbadian discovered that these filaments were actually an extension of the cell membrane, covered in proteins that aid in the transport of iron - cytochromes.
While microscopy enabled the researchers to understand the composition of the nanowire, they wanted to know if electrons could migrate along the cytochromes inside. A high density of these cytochromes might enable the electrons to move onto external surfaces.
Utilizing a technique called electron cryotomography, or ECT, the investigators could see the distribution of proteins that transport electrons in the nanowires. "These are not simple tubes," El-Naggar said. "They turned out to be more like a chain of membrane pearls, strung together."
While some of the transport proteins were touching, many were too far apart for an electron to traverse. The research team suggested that the proteins can float around the membrane. That would generate enough collisions to get electrons to move from one cytochrome to another until, at the end of the nanowire, they jump onto a rock or metal surface.
There is more work that remains to confirms that hypothesis. "My lab is driven by the idea that we could develop new machines, where living cells are functioning as part of a hybrid biotic-abiotic system," said El-Naggar. "We are trying [to] build the foundations of a new generation of living electronics."
Sources: AAAS/Eurekalert! Via USC, PNAS