Cleaning out Gut Bacteria may Improve Heart Failure
We have as many bacterial cells in our bodies as we do human cells, and those bacteria have a powerful impact on our health. Researchers have been exploring the many processes affected by the microbial community in our gastrointestinal tract, the gut microbiome. New findings have indicated that completely wiping the gut clean of bacteria could be beneficial for the heart and may slow damage that happens during heart failure.
It is thought that the microorganisms living in the gut increase the production of immune agents called T cells and thereby impact heart failure. In mice that model heart failure, antibiotic treatment over five weeks sterilized their guts and improved their outcomes. The findings have been presented at the 2018 meeting of Experimental Biology at the American Society for Investigative Pathology annual meeting.
"Our lab studies how the gut talks to the heart through T cells," said Francisco J. Carrillo-Salinas, Ph.D., a postdoctoral scholar at Tufts University who conducted the research. "Given that the gut is the body's largest reservoir of T cells and microbes, by modulating the microbiota we could modulate T cell activation and changes in the heart that lead to heart failure."
In heart failure, the heart can’t pump enough blood to the body. Almost 6 million people in the US have heart failure, and around half of those who are diagnosed die within five years.
Previous research has identified a link between the gut microbiome and cardiovascular health, as explained in the video.
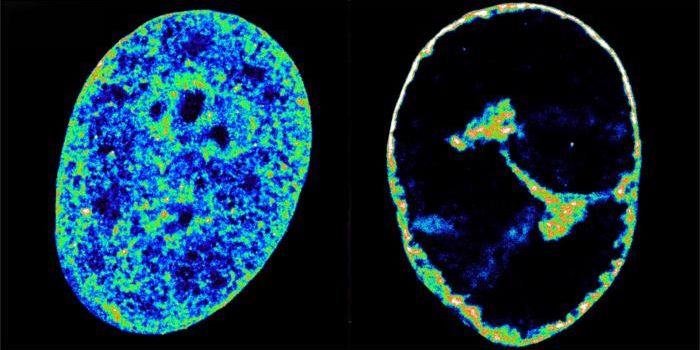
Carrillo-Salinas and collaborators have found that T cells penetrate into the heart in people that are experiencing heart failure.
Antibiotics are known to affect the gut microbiome, so researchers exposed mice modeling heart failure as well as normal mice to antibiotics, while some mice were untreated. The team assessed markers of heart function and immune activity in these mice.
Mice that both received antibiotics and modeled heart failure were better able to pump blood and had less tissue damage in their hearts compared to mice that did not get antibiotics.
"Because complete sterilization of the gut has proven to ameliorate some experimental models of T cell-mediated diseases, our results were in agreement with our initial hypothesis," explained Carrillo-Salinas. "The fact that we see fully preserved heart function is surprising, and I am looking forward to exciting new data on what happens in the heart once different bacteria recolonize the gut."
This work suggests that in the lymph nodes closest to the heart, T cells are activated, which then migrate to the heart and stop the progression of heart failure as they release cytokines. That enlarges the heart, and scar tissue is formed. In mice that had received antibiotics, none of these changes were observed.
"Understanding how the gut microbiota directly regulates the function of distant organs such as the heart will shed new light into potential new therapeutic approaches in patients recently diagnosed with heart failure to prevent progression," added Carrillo-Salinas. "Our results demonstrate that gut microbiota depletion prevents cardiac dysfunction and sets the stage for future studies that will determine which components of the microbiota are responsible for heart failure progression."
Sources: AAAS/Eurekalert! Via Experimental Biology 2018