Protecting Neurons Caught in the Danger Zone
Thanks to specialized cells called astrocytes, neurons are protected from an excessive immune response after brain injury so healthy neurons can begin the rebuilding process unaltered. Scientists are now researching how astrocytes receive their cue to jump into action; the rescue attempt they pursue is not in their normal arsenal of duties.
Following damage to the brain, either by trauma or a reduced blood supply, a cardiac condition called ischemia, the immune system springs into action. Damage to the brain in either case causes an influx of immune cells that sprout an inflammatory response that ends with a purging of all damaged neurons. The immune response can go overboard and harm healthy neurons as well, making an already harmful brain injury get much worse.
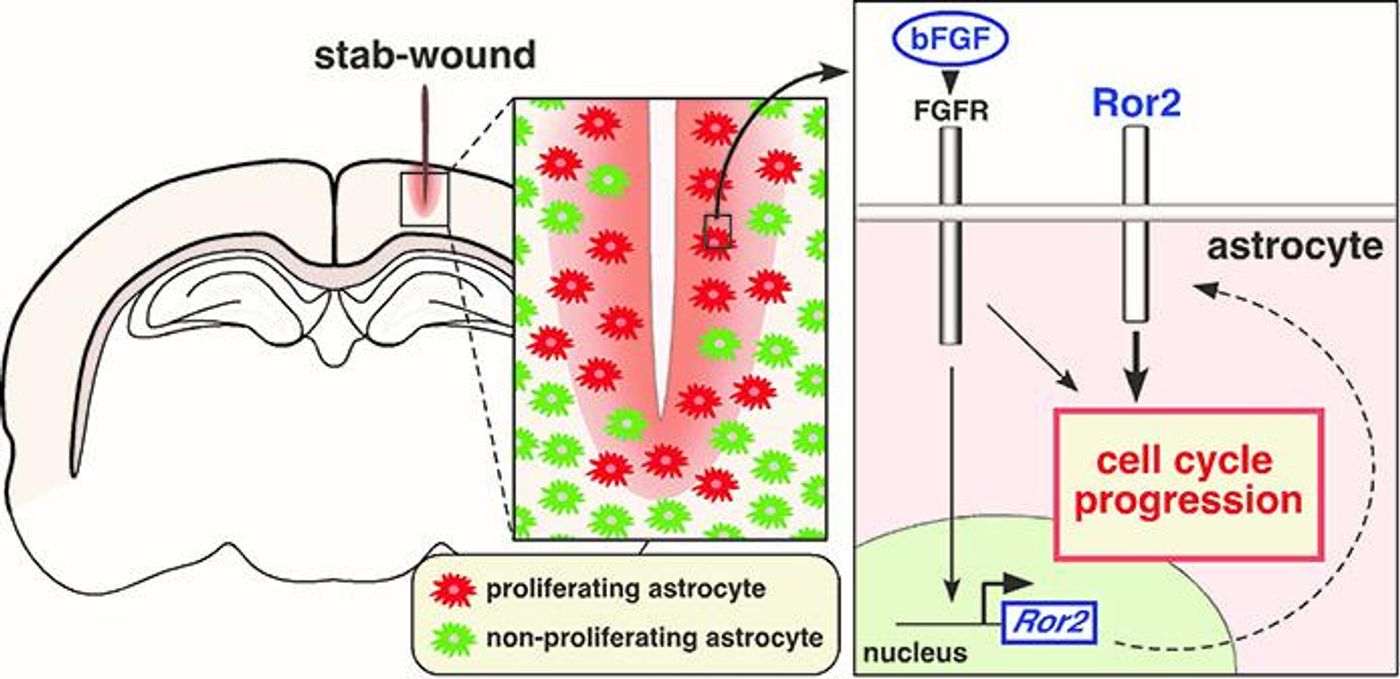
Astrocytes provide protection from an overbearing inflammatory response by surrounding damaged neurons and other cells caught in the crossfire during brain injury. However, researchers noticed that only specific groups of astrocytes were proliferating and protecting neurons and the select group of cells was only doing so in injured areas of the brain. How do the astrocytes know when and where to proliferate in response to brain injury?
From Kobe University, scientists found gene expression of receptor tyrosinase kinase Ror2 in select groups of astrocytes, a cell surface protein normally highly expressed only in neural stem cells. The same astrocytes that were found expression Ror2 were the cells proliferating and protecting neurons after brain injury, further underscoring a more complex connection.
Researchers tested their theory in mice models, with Ror2 gene expression inhibited in astrocytes in an experimental group. Compared to the control mice, Ror2 knockout mice showed a significant reduction in proliferating astrocytes after brain injury. In the middle of it all they also found basic fibroblast growth factor (bFGF), which seemed to be the middleman between Ror2 and astrocyte proliferation, as it activated the expression of Ror2 in the select group of astrocytes.
Only the astrocytes that responded to bFGF signals to express Ror2 would go on to proliferate and protect damaged neurons, and bFGF was produced by numerous cell types that escaped damage during injury, even neurons and astrocytes themselves.
Under the influence of bFGF and Ror2, neural-protective astrocytes were observed acquiring characteristics similar to neural stem cells. During normal conditions, astrocytes, the most numerous cell type in the human cerebral cortex, provide nutrition for neurons and regulate their activities.
These findings open all kinds of doors for new treatment options surrounding regeneration of neurons following brain damage by preventing excess damage to neurons in the first place. Something else to ponder: could the reduction of proliferating astrocytes after brain injury reduced during aging mean that the population of astrocytes expressing Ror2 are also decreasing? And is this connection related to an increased risk for senility and dementia during aging? Scientists will continue the search for answers to these questions and more. The present study was published in the journal Glia.
Source: Kobe University