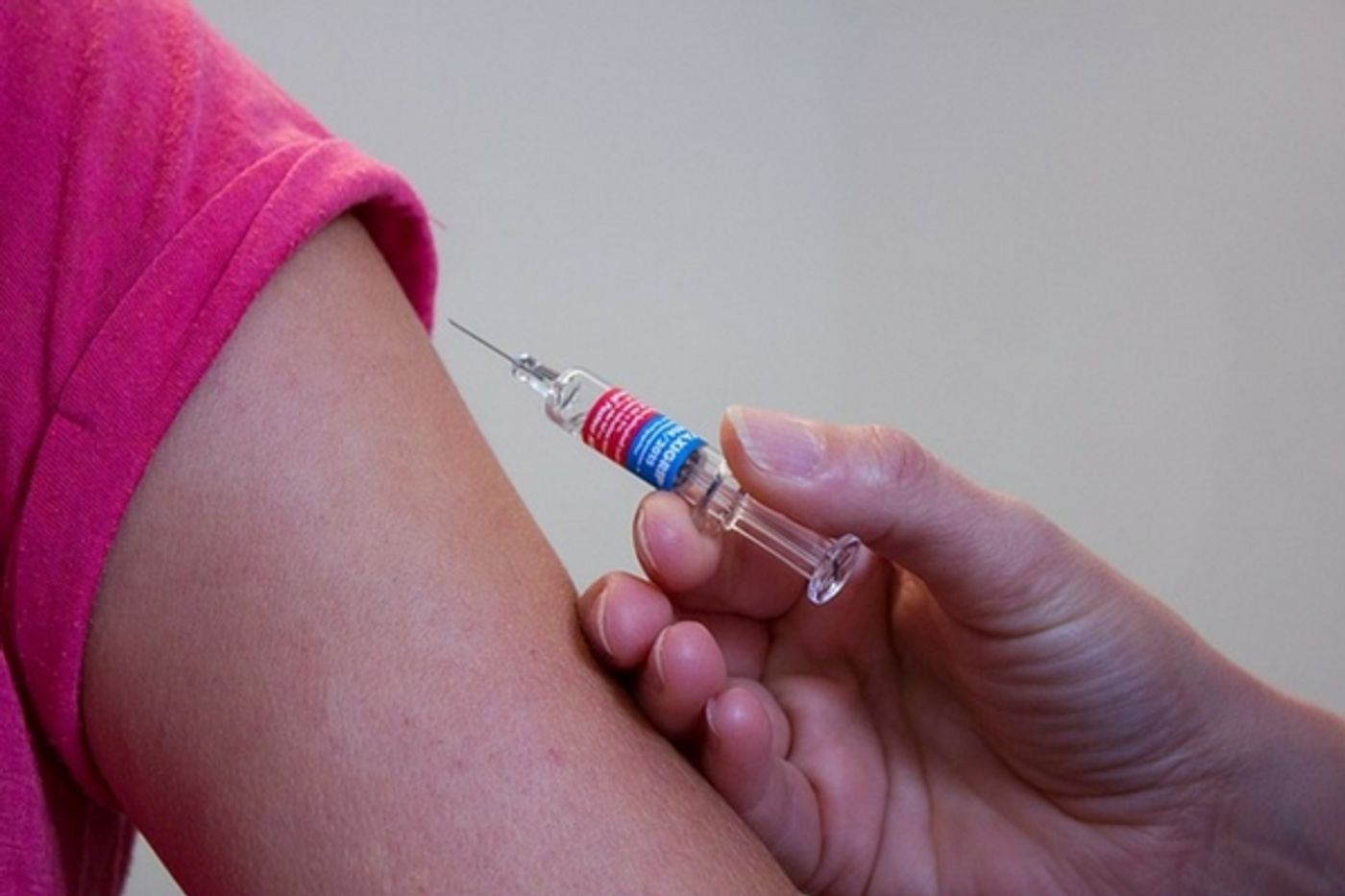
Zika Vaccine Trials Begin
While the Zika virus has not been in the headlines as much recently as when it was first discovered, there is still active work being done to combat the infection transmitted by mosquitos. Researchers from the National Institutes of Health are about to begin the first phase of human clinical trials for an experimental vaccine against the infection, which causes devastating neurological birth defects when contracted by pregnant women. The University of Miami Miller School of Medicine, along with other sites in the US and in other countries, is set to start full-scalle Zika vaccine trials this month.
The vaccine being tested is not like others, such as those that protect against measles or chicken pox by giving patients a small dose of the pathogen to trigger an immune response. Instead the Zika vaccine being tested is a DNA based vaccine. There will be no live or dead bits of the Zika virus contained in the shot. Instead it will contain small chunks of genetic material. Essentially it’s blank DNA that carries the protein building blocks of Zika in a long chain of pieces. While researchers have tested various DNA vaccines in mice, with great success, as yet, not one of them has worked in humans. Getting just the right sequence and type of the proteins that make up Zika is the key and it hasn’t happened yet, but the NIH researchers and scientists at the various test sites remain optimistic.
With the infection causing such severe neurological damage like microcephaly and impaired brain function, the chase is on to get it right. In addition to Miami, trials will happen in Houston at Baylor School of Medicine, and Ponce Health Sciences University in Puerto Rico. These three sites are considered Part A of the clinical trials and will focus on getting precise dosage amounts. Part B of the research will take place in cities outside the United States, including some in Mexico, Costa Rica, Brazil, Panama and Peru. These areas, known as “Zika Zones,” have seen high transmission rates of the disease. The trials in Part B will attempt to determine if the vaccine can actually prevent diseases and the accompanying neurological damage that are caused by a Zika infection. Study volunteers will be those from the area who have not had a Zika infection previously. The study will be double blind, where half of the participants will receive the vaccine and half will receive a placebo.
The numbers will start small with Part A enrolling 90 healthy men and non-pregnant women ages 18-35 years. The second phase, Part B will see enrollment jump to 2,400 men and non-pregnant women ages 15-35 years. The vaccine was developed by government scientists at the National Institute of Allergy and Infectious Diseases (NIAID) which is part of the NIH and the staff of researchers, doctors and scientists from the NIAID will oversee the program. The money for the program, initially thought to be in danger from the Trump administration budget cuts, is secure and costs for the research could reach $100 million.
The Vaccine Research Center (VRC) within the NIAID is responsible for the development version of the DNA based vaccine being tested. Early-stage human testing on the investigational drug was completed in 2016, after several rounds of animal testing before that. While Zika is a mild illness in most adults causing fever, joint pain and sometimes conjunctivitis, it can be catastrophic in pregnant women, causing their babies to be born with severe neurological damage and brain defects including microcephaly. Some studies have shown an association with Guillain-Barre syndrome, a condition that attacks the nervous system, but that link has not been proven conclusively. The trials are expected to be complete by 2019. The video below talks more about the historic trials which will soon begin, take a look.
Sources: University of Miami, via Eureka, STATNEWS.com, Science News