Mars' Crust May Affect the Planet's Atmospheric Composition
NASA has been exploring Mars since the 1960’s, but it wasn’t until today’s modern technology and advanced computers that they’ve been able to answer so many unanswered questions about the red planet. One of those is the abundance of certain isotopes in Mars’ atmosphere.
Using the Curiosity Rover, which first started crawling around on the red planet’s surface in 2012, NASA has been able to study the chemical content of Mars’ surface and atmosphere. In particular, they can use the on-board Sample Analysis at Mars (SAM) instruments to measure chemical composition and properties.

One of the more recent findings is the higher presence of krypton and xenon isotopes in the atmosphere than originally thought, both of which are “chemical variants” of the two elements because they have more neutrons than the basic form of the element.
It’s worth noting that these isotopes exist in greater amounts on Mars than they do on Earth, and scientists think they might know why.
In a study published in the Earth and Planetary Science Letters, NASA explains how Mars’ surface, the chemicals embedded in the crust themselves, could be contributing to the existence of these isotopes.
"What we found is that earlier studies of xenon and krypton only told part of the story," said Pamela Conrad, study lead author and SAM's deputy principal investigator at NASA's Goddard Space Flight Center. "SAM is now giving us the first complete in situ benchmark against which to compare meteorite measurements."
In essence, some of the isotopic existence may be a result of meteorites striking Mars, but that’s certainly not the only source of these isotopes.
Using static mass spectrometry, a way to analyze mere trace amounts of gases or isotopes, NASA was able to come up with the conclusions that some of the isotopes didn’t quite form as expected, suggesting there was more to the mystery than just meteorites.
The new theory comes by way of a natural process known as neutron capture, which is where the isotopes may be formed inside of the planet’s crust before being released into the atmosphere. Barium and bromine in the crust may have surrendered some of their neutrons to xenon and krypton respectively, producing what are known as xenon-124 and 126 and krypton-80 and 82.
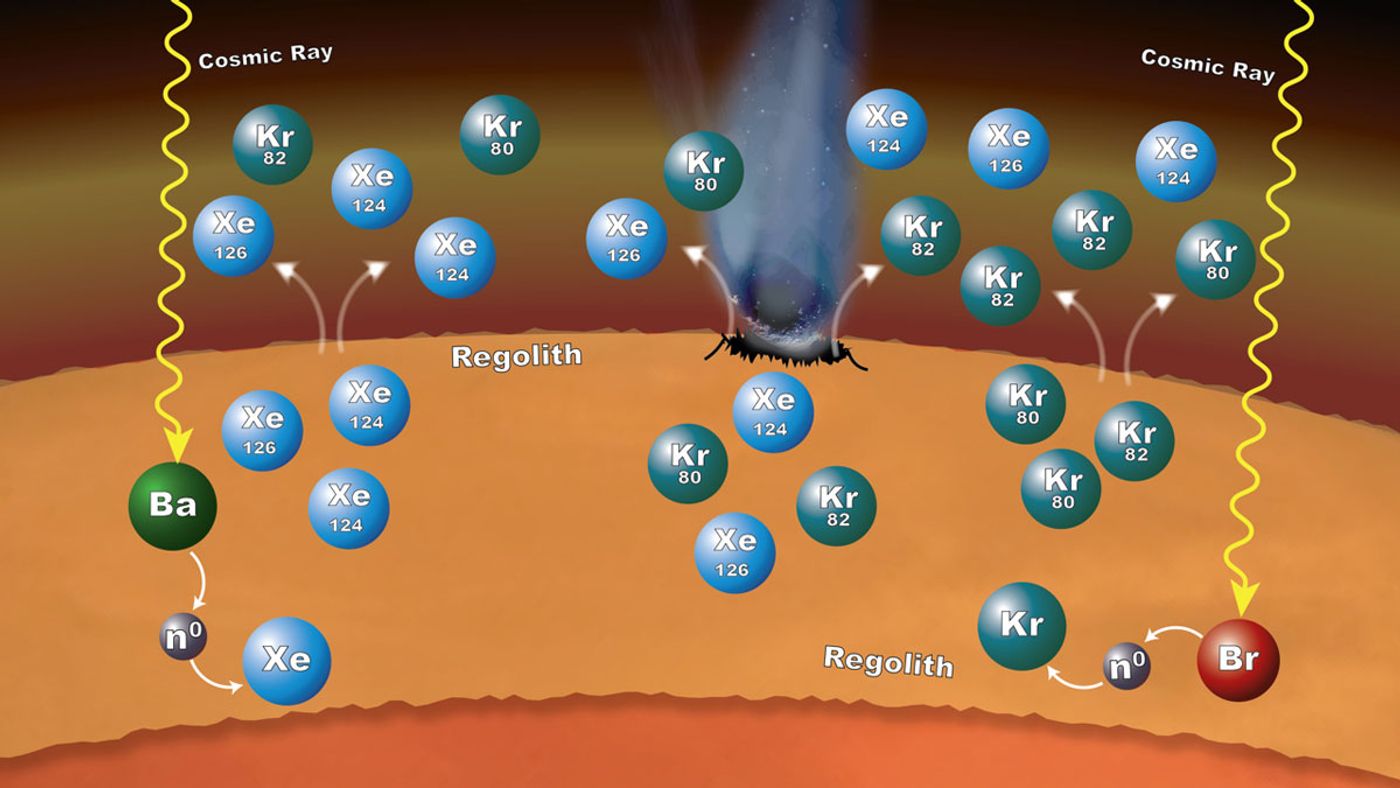
Image Credit: NASA/GSFC/JPL-Caltech
The impacts from space rocks may have helped release these gasses into the atmosphere, and over time, even the cracks in the red planet’s surface may have allowed them to escape the crust.
"SAM's measurements provide evidence of a really interesting process in which the rock and unconsolidated material at the planet's surface have contributed to the xenon and krypton isotopic composition of the atmosphere in a dynamic way," Conrad continued.
It's worth noting that the Mars 2020 rover is still under development and will, in coming years, become the latest edition of red planet-roving robots to study the planet, furthering our knowledge of its composition and the possibility that it may be able to support life. Equipped with the latest technology, it may even provide more clues into this study.
Source: NASA








