Why Breast Surgeons Are Calling for Wider Genetic Testing
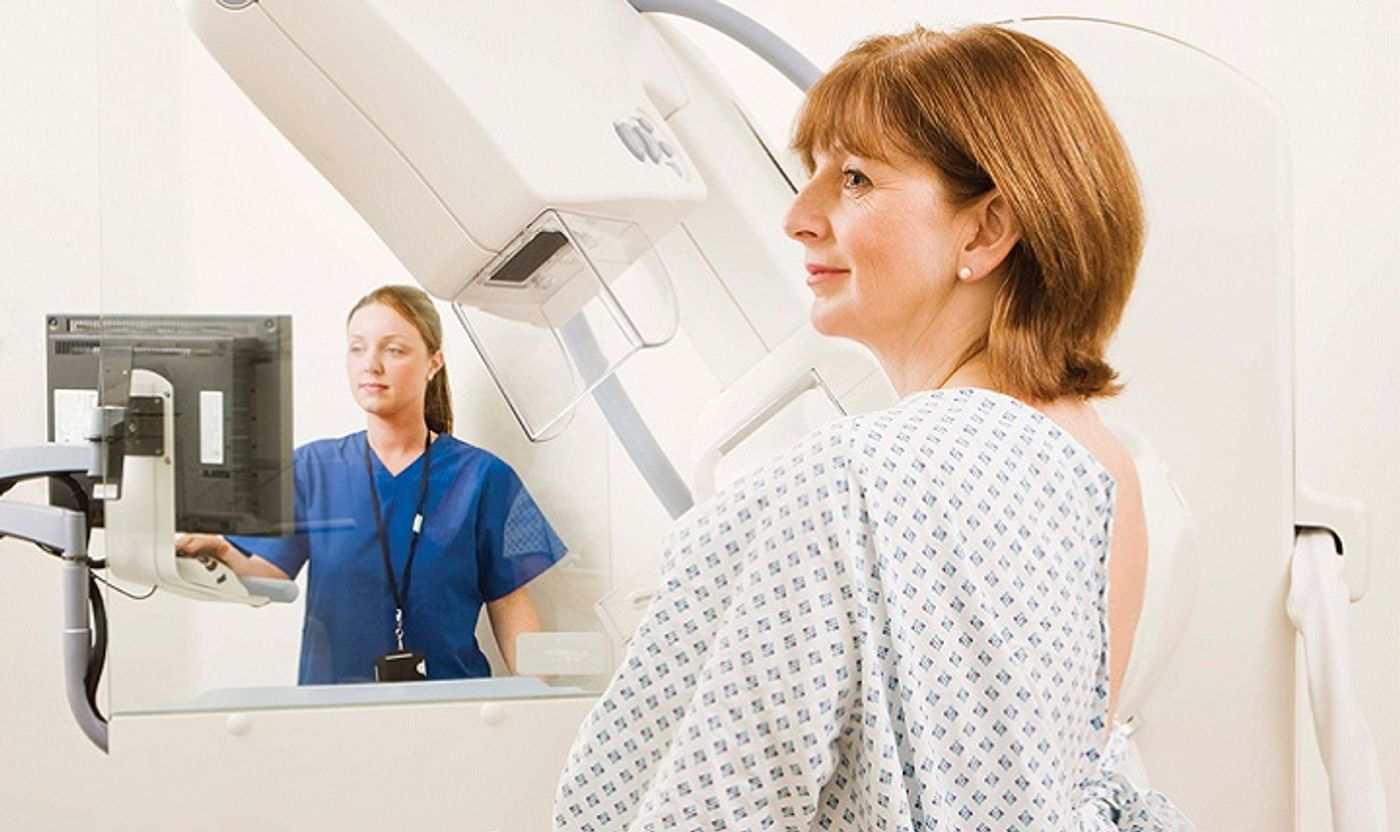
In early 2019, the American Society of Breast Surgeons (ASBrS) released new recommendations on genetic testing for people with breast cancer, widening the scope to include all such patients. This statement contrasts with the National Comprehensive Cancer Network’s (NCCN) more restrictive guidelines. ASBrS president and Texas Health Dallas surgeon Walton A. Taylor, M.D., says the NCCN guidelines are “fairly complicated” and hail from an era when testing was much more expensive.
“We are seeing more data that suggests the current NCCN guidelines miss a fair number of patients with genetic mutations: pathogenic variants that would change how we would manage their cancer, follow or screen them for future cancers, and what might happen to their children or siblings,” Taylor told Clinical Oncology.
The new ASBrS recommendations were developed by breast surgeons and medical oncologists with experience in genetics, risk assessment, health care policy and care management. The panel reviewed contemporary literature on genetic testing and concluded the NCCN guidelines were too limited.
In order to analyze the new guidelines, Dr. Peter Beitsch of the TME Breast Cancer Network in Dallas and his colleagues looked into the results of an 80-gene testing panel that was taken by breast cancer patients who had not previously received genetic testing. About half of the patients met NCCN criteria for the testing, and the other half didn’t. Beitsch and his team ran a broad panel test, looking not just for the about 30 genes associated with breast cancer, but the full 80 genes associated with cancers.
“[Not] surprisingly, the pathogenic variant rate is not statistically different between the two groups,” Beitsch said. Pathogenic or likely pathogenic variants were found in more than 9 percent of those who fell within NCCN guidelines, and almost 8 percent of those who did not. “With the current lower cost of testing, it doesn’t make sense for us to miss half the women who have a pathogenic variant,” he said, and his team concluded that all patients with breast cancer should have expanded panel testing.
Cost is one of the main concerns that arises with the potential expansion of testing. Most insurance only covers testing for patients who meet the NCCN guidelines. While the genetic test cost has now fallen to a few hundred dollars, it will still be a prohibitive amount to many patients.
“We’re hoping that by trying to move the needle we might persuade [insurers] to cover testing in an expanded fashion,” Taylor said.
Dr. Roshni Rao, chief of the breast surgery program and associate professor of surgery at New York-Presbyterian Hospital/Columbia University Medical Center, supports the new recommendations with some reservations, such as cost. She also points out the discovery of genetic abnormalities like variants of uncertain significance (VUS) can potentially cause overscreening and distress.
Establishing and maintaining a high-quality, transparently priced, connected and communicative network of genetic testing labs is another potential challenge related to expanded screening.
Sources: Clinical Oncology, American Society of Breast Surgeons