 UC San Francisco
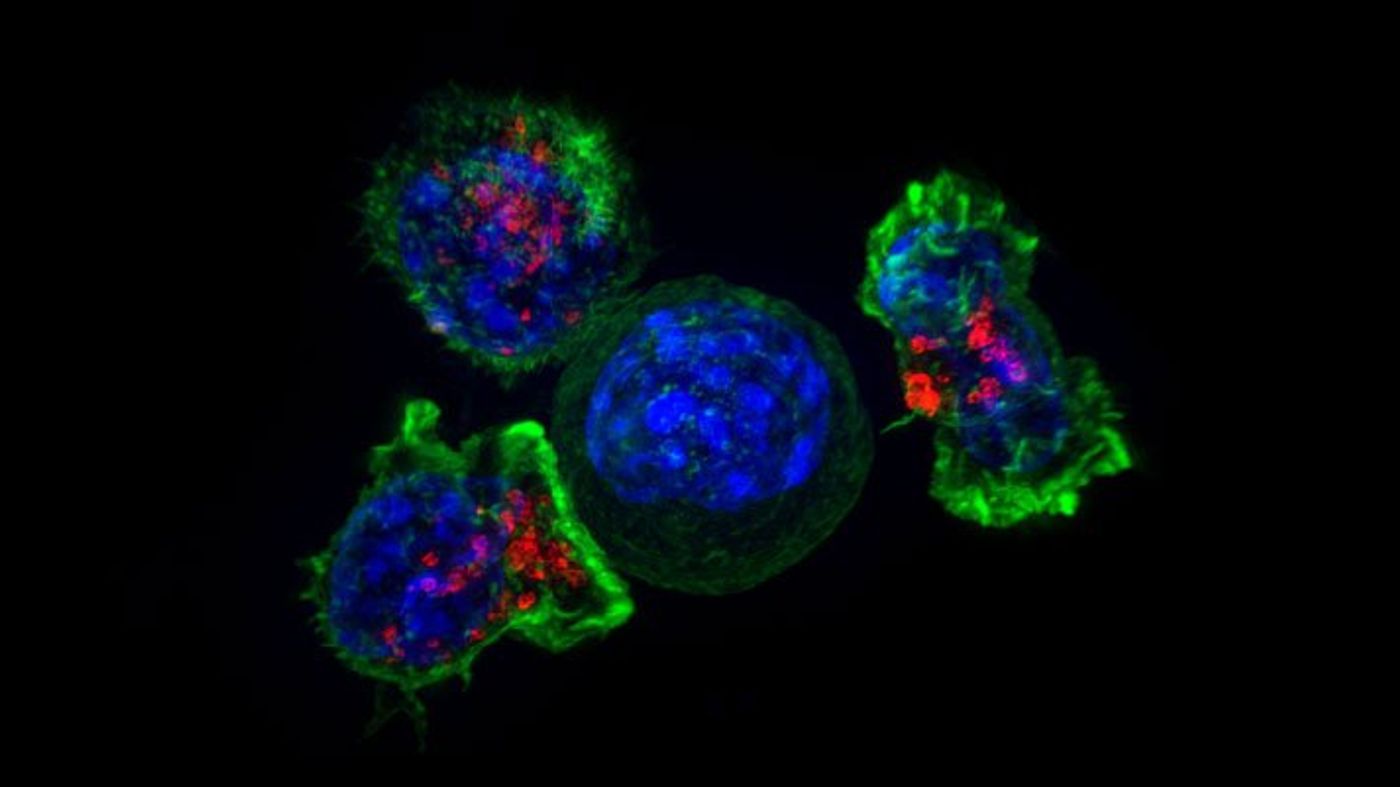
UC San Francisco scientists have developed a molecular “on switch” enabling tight control over the actions of T cells, immune system cells that have potential as cancer therapies. The innovation could sharply reduce severe, sometimes fatal side effects that have been a problem in advancing T cell-based treatments.
According to Wendell Lim, Ph.D., professor and chair of UCSF’s Department of Cellular and Molecular Pharmacology, and senior author of a new paper on the work, “Right now we put engineered T cells into patients and just hope for the best. This is the first of a series of ‘control knobs’ our lab is trying to create so doctors might have additional command over these cells once they’re inside the body.”
Scientists pursuing cell therapy have been refining cell-surface sensors called chimeric antigen receptors, or CARs. The CARs prompt T cells to zero in on particular proteins found primarily in tumors, where they launch cancer-killing immune responses.
CAR-equipped T cells can be successful in the treatment of chemotherapy-resistant leukemia, but they can cause side effects so severe that they may require monitoring in an Intensive Care Unit. Some patients have died after receiving CAR T cells or other engineered T cells.
Lim explained, “T cells are really powerful beasts, and they can be lethal when they’re activated. We’ve needed a remote control system that retains the power of these engineered T cells, but allows us to communicate specifically with them and manage them while they’re in the body.”
While some scientists have developed “suicide switches” that kill off CAR T cells if side effects become too dangerous, Lim thinks “that’s like shooting your own soldiers in the back. It requires completely aborting a complex and expensive treatment.”
As published in the September 24 issue of Science Express and reported in
Bioscience Technology, the UCSF team took the opposite approach, creating a new type of CAR T cell that is “off” by default. In the same way as conventional CAR T cells, according to the article, “These newly developed T cells will navigate toward and interact with cancer cells, but will not launch any immune assault unless a specially designed drug has been administered. This controller drug forms a chemical bridge between components inside the CAR T cells, flipping the cells into an active, on status. When the drug is no longer present, the T cells revert to an off position.”
Lim concluded, “That we can engineer CAR T cells to have slightly different, quite powerful effects – even if for a subset of patients or for certain types of cancer – is really remarkable, and this is just the tip of the iceberg.”
 UC San Francisco
UC San Francisco