"Saturating" the Liver with Chemotherapy to Treat Cancer
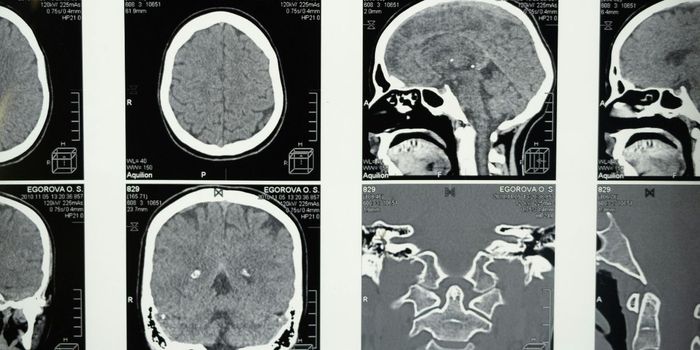
Uveal melanoma is a rare type of cancer that grows in the melanin, the dark-colored pigment of the eye. About half of patients with uveal melanoma experience metastasis characterized by the spread of cancer cells to distant parts of the body. The liver is the most commonly inflicted secondary organ in metastatic uveal melanoma (mUM). Sadly, 75 - 90% of mUM cases prove fatal.
Currently, there is no standard-of-care treatment for mUM and developing an efficacious treatment modality remains a research focus. Immunotherapies are used in some cases, but they garner less than a 20% response rate in treating mUM. Percutaneous hepatic perfusion of melphalan (M-PHP) has emerged as a promising investigative technique for treating liver metastasis. M-PHP has many benefits, including that it is a minimally invasive procedure that typically results in minimal adverse effects. Initial trials suggest M-PHP could significantly benefit patients with liver metastases.
M-PHP administers chemotherapy directly to the liver to treat liver cancer or liver metastases. Because the liver is soaked, or “saturated”, with high-dose chemotherapy, this technique is considered “chemosaturation” therapy.
Doctors insert a thin tube, known as a cannula, into the hepatic artery, the blood vessel that delivers oxygenated blood to the liver and pancreas. Melphalan, a chemotherapy, is then delivered to the liver via the cannula. To limit damage to healthy tissue throughout the body, doctors will perform a venobenous bypass procedure. The chemotherapy-infused blood exiting the liver is collected, the harmful chemotherapeutic metabolites are filtered out, and the blood is returned to the patient, re-entering normal circulation.
To expand on some of the clinical data on M-PHP, a group of researchers from the United Kingdom initiated a retrospective study analyzing the outcome of a large cohort of patients. The study results, recently published in Melanoma Research, demonstrate safety and efficacy for mUM patients with liver metastasis treated with chemosaturation therapy.
The researchers utilized data from 81 mUM patients who received M-PHP. On average, patients underwent three rounds of treatment. The authors reported the disease control rate at 88.9% and the overall response rate at 60.5%. At the time of analysis, the median follow-up time was about 13 months. The study found that the median overall progression-free was 8.4 months, and the median overall survival was almost 15 months.
The authors acknowledged some limitations of their study, including the lack of a control or alternatively treated arm. Further, the authors suggest that combining chemosaturation with immunotherapy could further enhance the benefit to mUM patients. In particular, they indicate IDO and TIGIT as potential targets for a combinatorial immunotherapy approach.
Sources: Oncol Ther, Opthamology, Oncoimmunology, Hepatoma Res, Ann Surg Oncol, Mel Res, Transplantation Proc, Cancer Cell