As the patent protection on the blockbuster anticancer drug Gleevec expired earlier this year, the now available generic version is expected to work just as well while saving leukemia patients and insurers
thousands of dollars. In an era of ever-increasing healthcare costs, affordable generic alternatives are a welcome relief for patients, especially those who require life-long, daily drug interventions.
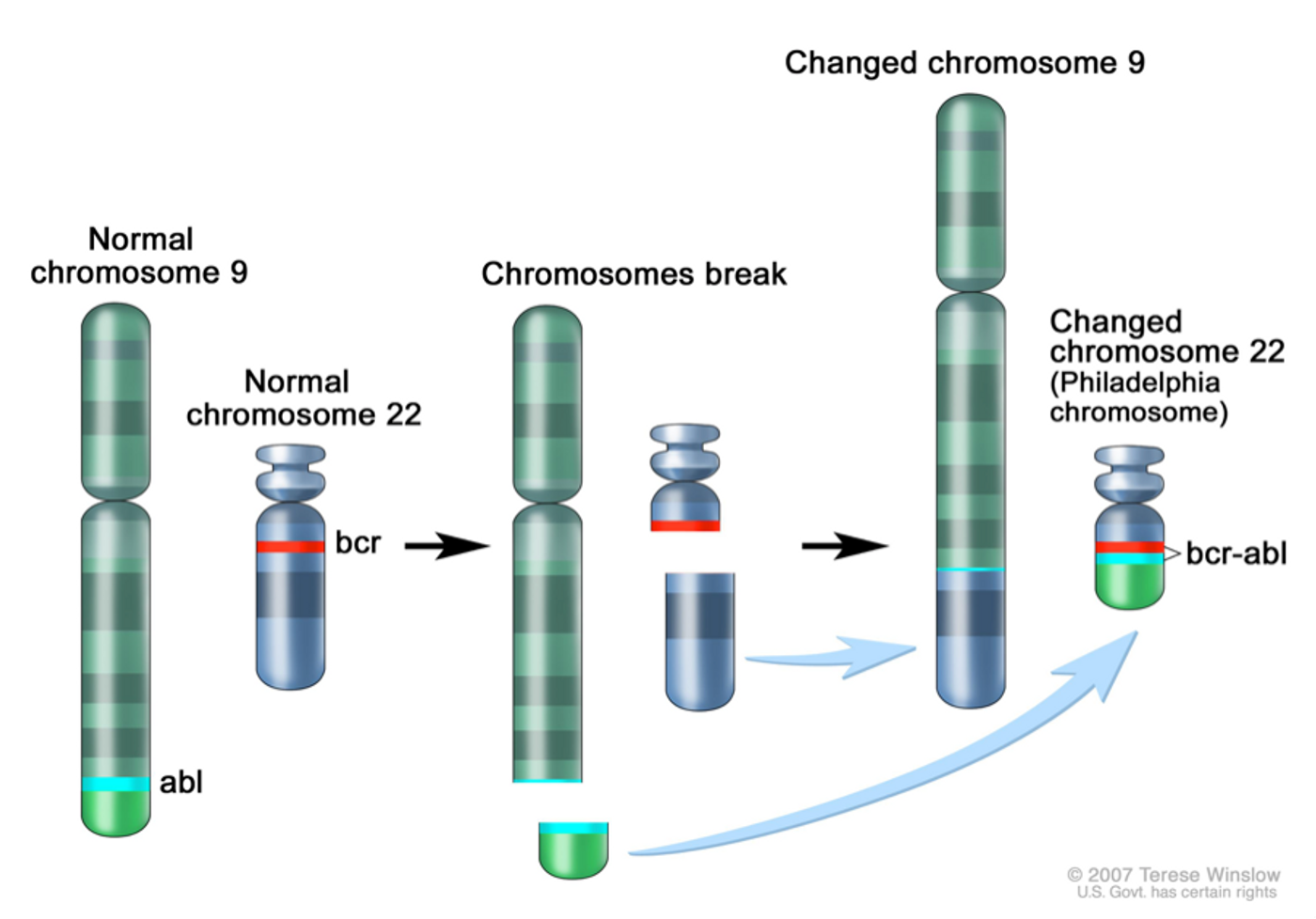
Chronic myeloid leukemia (CML) is a deadly cancer of the white blood cell that is associated with a well-known abnormal chromosomal event that swaps the ends of two chromosomes. The resulting translocated Philadelphia chromosome contains sequences for a fusion protein known as BRC-ABL1. This mutant protein is a tyrosine kinase that is always activated and thus causes the uncontrolled growth of myeloid cells in the bone marrow, leading to CML.
Tyrosine kinase inhibitors, like
Gleevec (Novartis), work to turn off the activity of these kinase enzymes, which stop cancer cells from proliferating. When it came on the market in 2001, Gleevec completely transformed treatment for CML patients. No longer was the CML diagnosis considered an inevitable death sentence; Gleevec made CML a manageable disease with 89% five-year survival rates.
But surviving CML comes at a huge financial cost of around $75,000 for every year’s worth of Gleevec supply. By contrast, the generic version of Gleevec,
imatinib, could cost less than $6,000 a year. This estimation is based on published cost data for newly diagnosed CML patients and real-world savings from Canada, where the generic imatinib has been prescribed as a first-line treatment for CML in the past year.
Cost-analysis by researchers at the Johns Hopkins Bloomberg School of Public Health suggest that the U.S. health system could stand to save $100,000 per patient over five years. By extension, for every 100 CML patients on the generic version for five years, one insurance company alone could save $9.1 million.
"There is minimal risk to starting all patients on imatinib first," said William Padula, assistant professor at the Bloomberg School and first study author. "If the patient can't tolerate the medication or it seems to be ineffective in that patient, then we can switch the patient to a more expensive drug. Insurance companies have the ability to dictate which drugs physicians prescribe first, and they regularly do. Doing so here would mean very little risk to health and a lot of cost savings."
An obvious question arising from such results is why brand name drugs, like Gleevec, cost so much when the generics cost significantly less. The discrepancy in such pricing is partly why the pharmaceutical industry is often demonized for exacting huge costs on life-saving drugs. In response, ‘big pharma’ has always countered with the laborious research and development efforts required in getting a drug through the testing pipeline. Indeed, a study by the Tufts Center for the Study of Drug Development estimated that it costs a mind-numbing $2.6 billion to get a drug from an idea to a marketable reality.
Still, Padula argues that, unlike cars, the prices of drugs have little to do with how much it costs to make the final product. "When patent protection is lost, the prices are set closer to the true cost of the drug," Padula added. "They're making 'General Motors' profits as opposed to Pharma profits and that savings can be shared with the consumer, but only if doctors and insurers work together to make sure patients are being prescribed the more cost-effective medication."
Additional source:
EurkekAlert!