Enzymes Form a Molecular Switch Vital to Cell Division
Cell division is one of the most fundamental processes in biology. In eukaryotes, there are several phases of the cycle, during which DNA is replicated and the cell divides in two. You can see a visual explanation of the cycle in the video below. One phase is called G1, and the transition from it to the next phase, S, is not well understood. Publishing in Cell Reports, scientists at the University of North Carolina School of Medicine have found that the boundary between the phases is controlled by a molecular switch, made up of two enzyme complexes.
This finding may have implications for the development of cancer drugs in the future; the enzyme interaction might be one drug target, ideally halting the uncontrolled division that is a hallmark of cancer cells.
"Our work describes a key piece of the puzzle for how this fundamental process works," commented senior author of the study Michael J. Emanuele, PhD, an Assistant Professor in Department of Pharmacology, and a member of the UNC Lineberger Comprehensive Cancer Center. "We did not focus on suppressing cancer cell division in this study, but given what we've found we think it's definitely worth investigating further."
In the G1 phase cells ramp up the production of key molecules and proteins to populate the new daughter cell. When cells move from G1 to the S phase, there’s no coming back; from there division must continue or the cell dies trying. In S phase, chromosomes are duplicated and then separated.
The complex gating the boundary is called APC/C, composed of over a dozen different proteins.
While it was previously found that APC/C works in collaboration with a protein called Cdh1 to collect and tag proteins for destruction, a common cellular mechanism for regulating protein levels. In this case, the APC/C complex is controlling the levels of proteins that help move the cell into the next, S phase; while APC/C is active, the cell is prevented from moving into S phase.
"Shutting off APC/C is essential for cells to get across the G1/S border. We've known that for a really long time, and we've also known that this requirement exists in the cells of all organisms from yeast to humans,” explained Emanuele. “But just how APC/C is shut off to permit this border crossing has not been clear."
Another molecular mechanism for tagging and disposing proteins involves a complex called E3 ubiquitin ligase. Emanuele's team discovered that APC/C and Cdh1 act on a protein called cyclin F, which functions to enable a E3 ubiquitin ligase called SCF to recognize its targets. As such, one large ubiquitin ligase complex, APC/C-Cdh1, works to halt the action of SCF-cyclin F. The team then showed that SCF-cyclin F works on the APC/C-Cdh1 complex in turn.
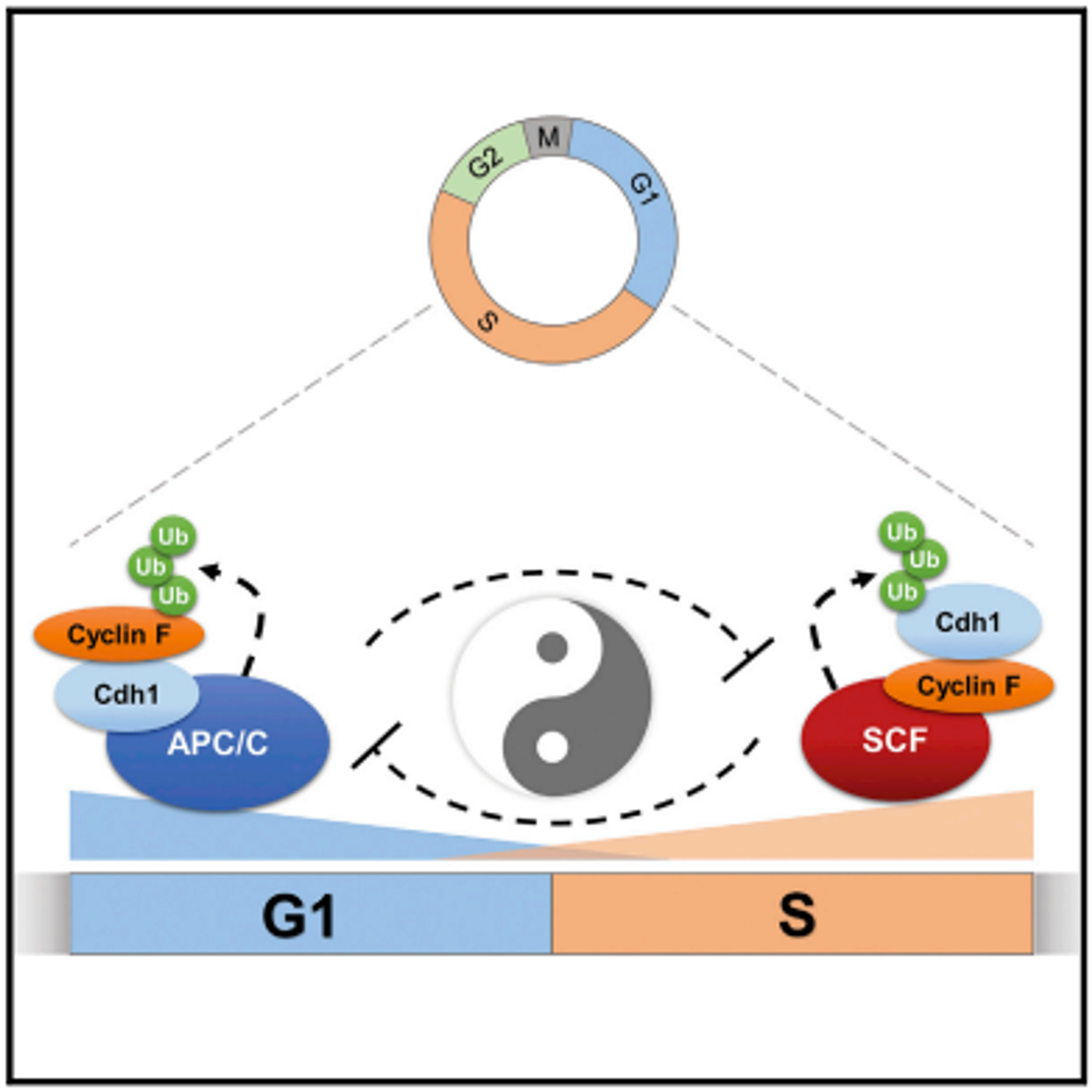
"When we looked at cells going through the cell cycle, we saw cyclin F levels starting to increase right at the G1/S transition, and at the same time the levels of Cdh1 were decreasing. The timing was perfect," Emanuele explained. "Then, when we put the protein complexes in cells at the same time, the levels of both went down, as if they were targeting each other."
Additional experiments confirmed that APC/C-Cdh1 functions to target cyclin F and effectively shut down SCF/cyclin F, the action is reciprocated as Cdh1 turns off APC/C-Cdh1. That second switch moves the cell into S phase.
"To the best of our knowledge nobody else has described a system involving direct antagonism between two E3 ubiquitin ligases," said Emanuele. "It suggests the possibility that similar mutually antagonistic pairs of E3s regulate other oscillating systems in biology."
The exact details of the switch - how APC/C prevents the cell from moving into S phase and how SCF/cyclin F overpower APC/C to start S phase - remain unknown. It is suspected by Emanuele that a common modification to proteins such as phosphorylation may be a part of the mechanism. "That's something we're working on now," he added.
This research may help the understanding of uncontrolled cell division and other unknowns of the cell cycle. “Cdh1 is a largely under-appreciated tumor suppressor. Although it blocks S phase, Cdh1 isn't mutated in cancers. One of the implications of our data, however, is that Cdh1 is degraded at this critical G1/S juncture. It could be that in some cases, too much Cdh1degradation is all that's needed to promote cancerous growth,” Emanuele concluded.
If you would like to know more about how the cell cycle gets out of control in cancer, watch the video above.
Source: Science Daily via UNC, Cell Reports