Scientists Attack the Telomeres to Halt Brain Cancer
By targeting the endcaps of cancer cell chromosomes, researchers may have landed on a promising new therapy to treat the notoriously lethal brain cancer known as glioblastoma.
Glioblastoma is often nicknamed “octopus tumor” because the malignant glial cells form vast networks of tendrils throughout the brain. This makes it extremely difficult to completely get rid of the tumor. As such, the brain cancer can evade even the most aggressive surgeries, chemotherapies, and radiotherapies, leaving patients with a five-year survival rate of less than 10 percent. Over 70 percent of patients diagnosed with this type of brain cancer rarely survive past the two-year mark.
One mechanism that allows these tumors to infiltrate the brain so rapidly is the presence of cells called glioblastoma stem cells (GSCs). These cells confer the ability for the cancer to self-renew and affords protection from different therapies. In essence, GSCs help the tumor penetrate deeper into the brain.
When the team from the Spanish National Cancer Research Centre (CNIO) in Madrid took an interest in glioblastoma, they already had a focus in mind. That is, they hoped to find the cancer’s weakness in its telomeres.
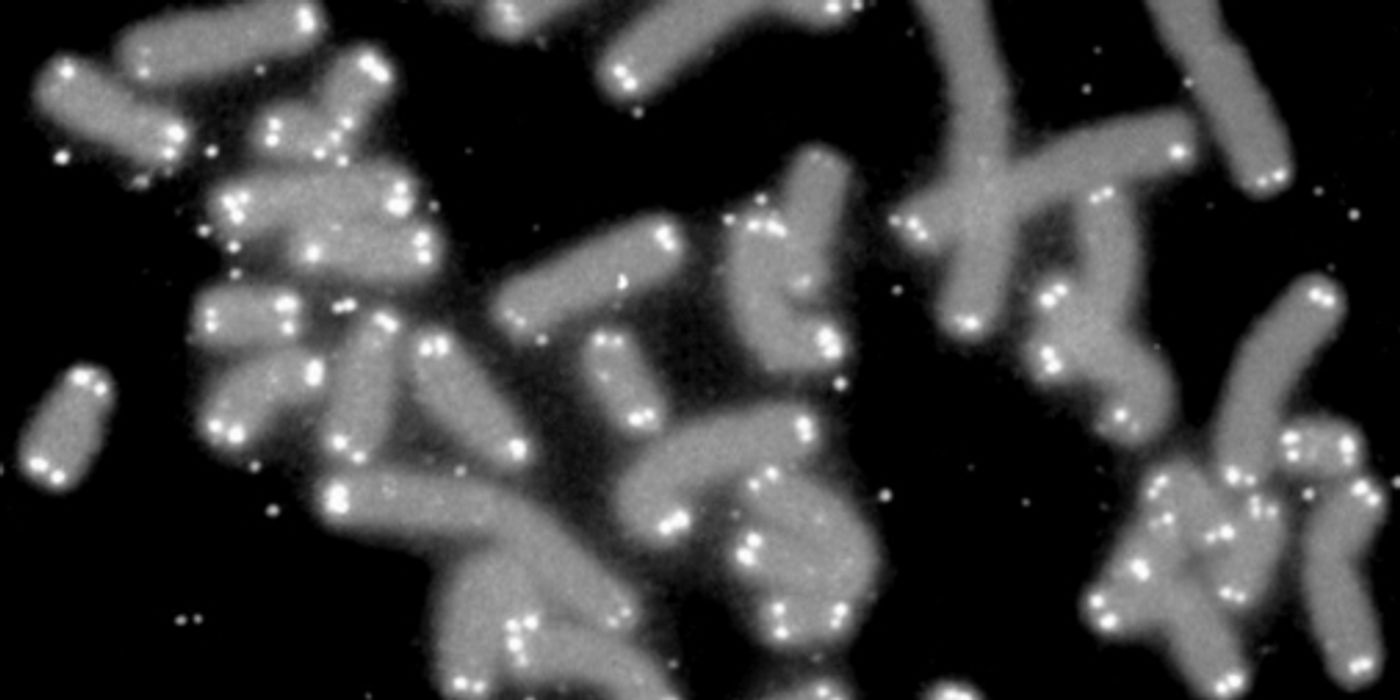
Telomeres are protective caps at the end of our chromosomes. These structures get shorter and shorter with each successive cell division. And when the telomeres get to a critical short length, the cell is signaled to die. In this way, telomeres act as a built-in timer for cell death.
When the team scrutinized GSCs, they found an abnormal amount of a protein called Telomeric Repeat Factor 1 (TRF1). The high level of this protein likely enables the cancer telomeres to escape the usual shortening and divide past their expiry status.
When TRF1 was knocked out in mouse models of glioblastomas, researchers saw dramatic reduction in tumor size and 80 percent increase in survival. When human glioblastoma cells were transplanted into mice, treatment of TRF1 inhibitors yielded similar results: tumor size reduction and longer survival time.
"It has a major therapeutic effect on glioblastoma. We see that inhibiting TRF1 is an effective strategy for treating glioblastoma both by itself and in combination with current radiation and temozolomide therapies,” said Dr. Maria Blasco, the study’s senior author, and head of the Telomeres and Telomerase Group at the CNIO.
Of note, when the team blocked TRF1 in mice with already formed glioblastoma tumors, the survival was 30 percent as compared to 80 percent. This suggests the treatment may not be as effective in the later stages of tumor development.
Next, the team will test different TRF1 inhibitors and in conjunction with other chemotherapeutic agents. The goal is to identify a combination that will surpass the outcome of a current chemotherapy (temozolomide) and radiation regimen, which gives patients a median survival length shy of just 15 months.
Additional sources: MNT