A DNA based nanorobot to search and destroy cancer!
Science fiction film Fantastic Voyage is a well-known motion picture where miniaturized nano-submarine is injected into a comatose scientist to search and destroy his brain clot. This kind of nanomedicine may not be possible, but scientists have created a nano-sized DNA origami which transforms after encountering tumor and kill it using its payload.
DNA based origami robots have been shown to sense the environment and actuate a function. However, targeted in-vivo delivery of a drug using DNA origami-based robots has never been shown until now. A team of Chinese scientists has recently published a paper in a peer-reviewed journal of Nature Biotechnology, which describes DNA based robots for targeted controlled delivery of cancer-destroying chemical in-vivo.
Most of the cancers are solid tumors which are known to rely on newly developed blood vessels for the continuous source of nutrients and oxygen for the growth of the tumor mass. Targeting these blood vessels by occlusion can initiate a cascade of the cataclysm of cells death within the tumor. Occlusion of tumor blood vessel leads to thrombus formation which is very difficult for tumor cells to overcome in the form of tumor resistance. Moreover, occlusion based therapy can be used for many different types of tumors as all of them rely on blood vessel formation.
Thrombin is a type of protease which activates platelets and form obstructive thrombosis. However, naked thrombin is short-lived and has off-site target side effects, and that is why its use is limited in cancer therapeutics. In the present paper, the team of scientists used DNA origami to protect thrombin from the harsh in-vivo denaturing environment. The release of the thrombin from DNA nanorobots was triggered only by tumor blood vessel marker, nucleolin.
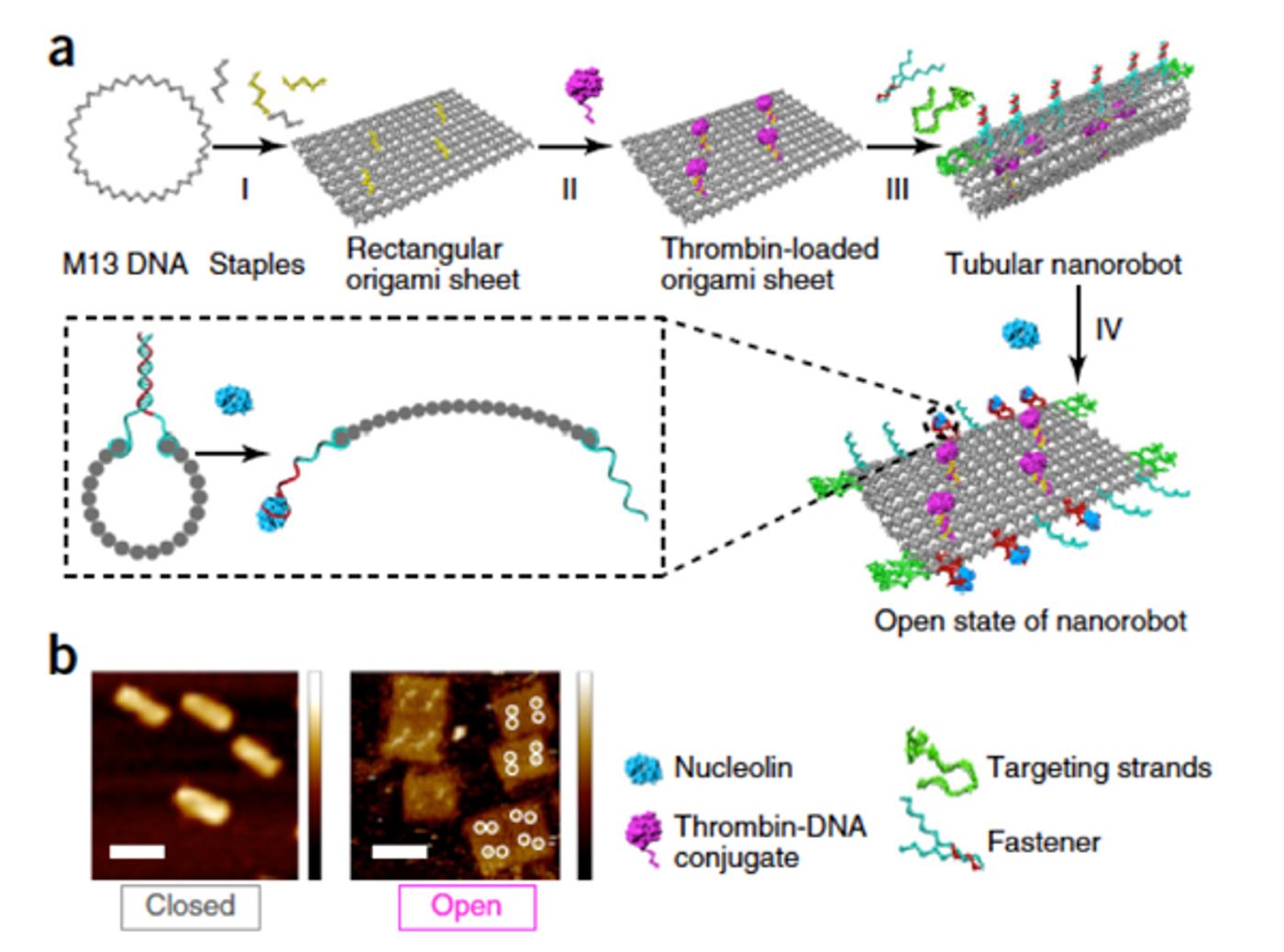
First, the team used a single-stranded M13 bacteriophage DNA to make a rectangular origami sheet which was then chemically anchored with thrombin at specific sites. They did in-vitro biological tests to make sure thrombin enzyme is active within the nanorobots. Using DNA aptamers as the fasteners, they made rectangular sheet into tubular structure to surround thrombin within the hollow tube to protect against plasma fibrinogen and platelets. DNA aptamers are artificially made small oligonucleotides which can be designed to bind a specific molecule. Here in this study, scientists used DNA aptamers which could specifically bind nucleolin present on tumor blood vessels. Many in-vitro tests conducted by them showed aptamer fastener could recognize nucleolin present on actively proliferating endothelial cells of the tumor. Upon binding, aptamer reconfigured and allowed tubular structure to deliver its cargo, thrombin at the binding site. Furthermore, they used a mouse model of melanoma to show the therapeutic potential of their nanorobots. The data from those experiments shows the occlusion of the blood vessel at tumor site leading to reduced tumor burden and statistically significant survival in treated mice. In this melanoma model, DNA nanorobots not only reduced primary tumor but also decreased the metastatic score.
Note: DNA origami as nanostructure; Courtsey: iBiology
Finally, authors suggest that this new technology in future can be used as a drug delivery mechanism for other diseases by changing the targeting moieties and loaded drug molecule.