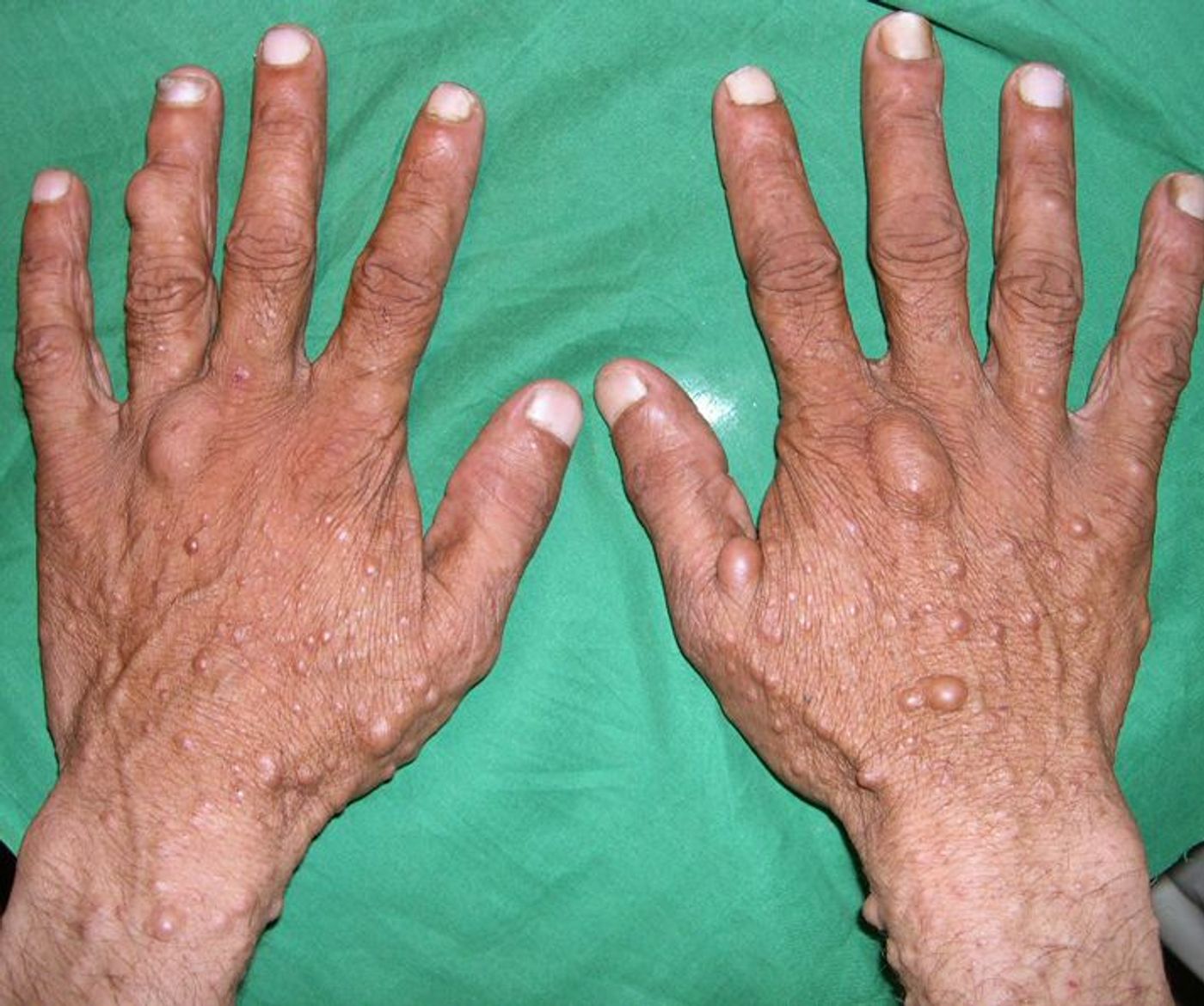
Neurofibromin is an important protein found in all of your cells that naturally suppresses tumor formation and is an anti-cell proliferator. It also inhibits Ras proteins, also found in all of your cells, which stimulate cell growth, differentiation, and survival. When neurofibromin is missing due to a genetic mutation, multiple complications arise.
You get one NF1 gene from your mother and one from your father. Depending on whether one or both of these genes are mutated, a relatively common occurence, a child can be born with varying severity of
neurofibromatosis, one complication of which leads to premature cardiovascular disease. Out of 50-70 thousand NF1 patients, about seven percent develop cardiovascular complications. Others develop cancer, bone disease, and cognitive issues.
While this gene mutation is inheritable, the mutation can also be acquired spontaneously, which occurs in 30-50% of individuals with neurofibromatosis.
From the Medical College of Georgia, scientists like Dr. Brian Stansfield are studying two different cardiovascular complications that arise from cases of neurofibromatosis, and their effects are practically opposite.
Arterial stenosis occurs when blood flow is hindered by a loss of flexibility in the artery walls, potentially leading to hypertension and stroke. On the other hand, neurofibromatosis can also lead to aortic aneurysms, where patients are excessively vulnerable to internal bleeding from weak vessel walls.
Dubbed "cancer of the cardiovascular system" by Stansfield, NF1 gene mutations disastrously wreak havoc in the body when the NF1/Ras pathway can't be regulated effectively. Stansfield and his team looked to investigate how the two cardiovascular complications of neurofibromatosis are caused by the same initial genetic mutation, and what makes one condition occur instead of the other. By exploring the pathology behind both of these conditions, the team from Georgia hopes to provide better diagnosis and treatment of young, high-risk patients.
In their study, the team used animal models of both arterial stenosis and aortic aneurysms. The NF1 mutant models of arterial stenosis showed an increase of macrophages in affected arteries. These lymphocytes deliver more growth factors, in addition to Ras proteins promoting cell growth, uninhibited due to the absence of required neurofibromin. In the models of aortic aneurysms, the scientists observed an increase in matrix metalloproteinases, enzymes that break down cell receptors and stimulate cell death, leading to a weakening of artery wall integrity.
Understanding the implications of disrupting the NF1/Ras pathway is vital to the future treatment of neurofibromatosis patients. Stansfield and his team now have plenty of targets (macrophages, Ras proteins, matrix metallophases) as options for manipulating cell dynamics in order to treat patients.
Watch the following to learn more about the general impact of neurofibromatosis.
Source: Medical College of Georgia,
JagWIRE