Reversing the Heart Damage Seen in Marfan Syndrome
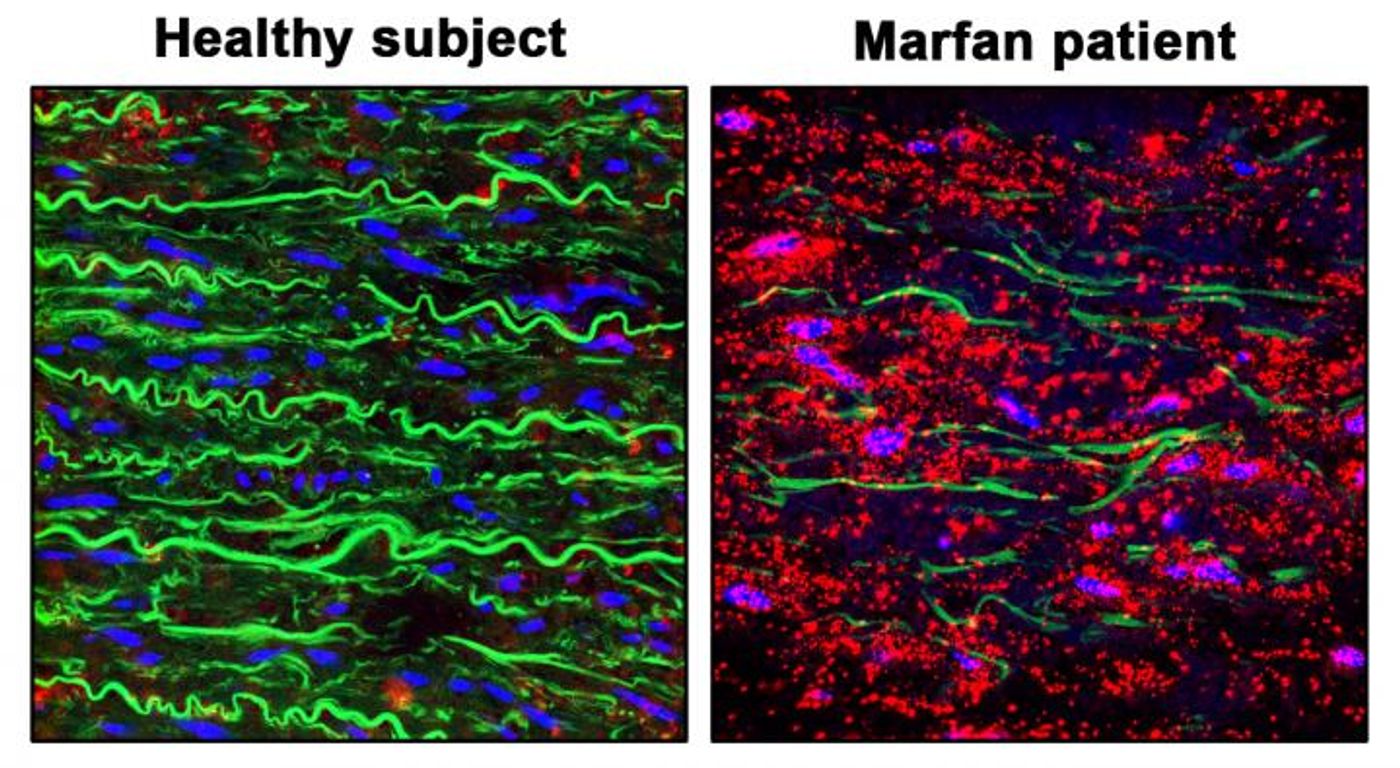
Marfan syndrome is a genetic disorder that disrupts the connective tissues that anchor and link the body's organs, affecting the eyes, heart, skeleton, and blood vessels. Patients with Marfan often experience a progressive weakening in the aortic wall of the heart, called aortic aneurysm (AA). While AA is not always a problem, it can cause the aorta to rupture or dissect. This complication, called thoracic aortic aneurysm and dissection (TAAD) is also common, a lethal condition.
Researchers have now found that a biochemical pathway involving nitric oxide (NO) is overactive in the aortas of both Marfan patients and a mouse model of the disease. This overactivation seems to be the cause of aortic aneurysms in Marfan patients. These findings have been reported in Nature Communications.
"Aortic dissection accounts for more than 90 percent of deaths associated with Marfan Syndrome," noted the co-director of the study, Dr. Juan Miguel Redondo of Centro Nacional de Investigaciones Cardiovasculares (CNIC).
Right now, treatments for Marfan syndrome aim to reduce the pressure blood exerts on the arterial wall, but do not stop the wall from degenerating. Surgery is the only option for patients with aortic disease. Now that NO has been identified as a crucial player in the aortic disease seen in Marfan syndrome, the NO pathway can be investigated for its potential as a biomarker or drug target.
The new study has built on other observations by this research team. "We previously detected high expression of a protein with a high capacity for NO production in the aortas of Marfan Syndrome patients and an animal model of the disease, and we therefore undertook an in-depth investigation of the role of NO in the associated aortic disease," explained study co-director Dr. Miguel Campanero of the Centro de Biología Molecular Severo Ochoa (CBM-CSIC-UAM).
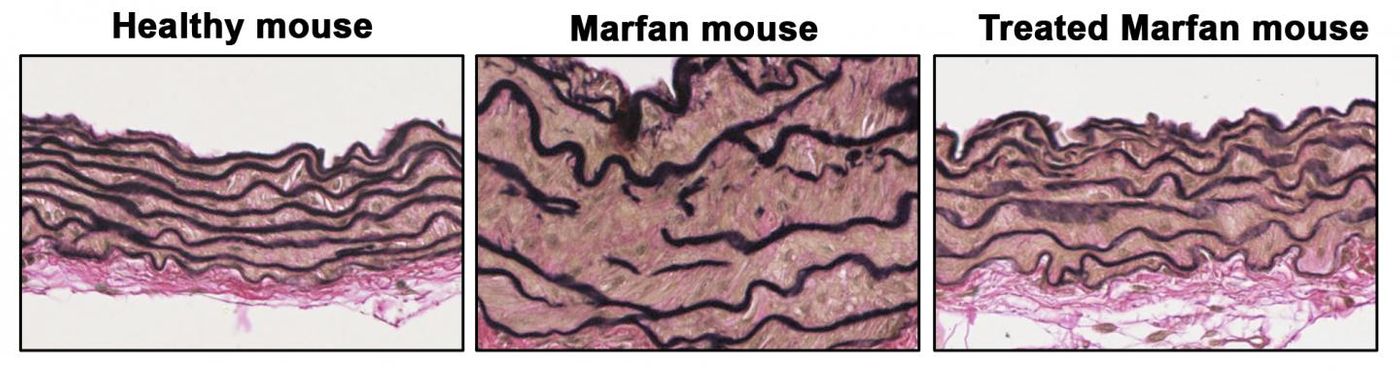
The scientists found that in Marfan Syndrome, high levels of NO can lead to TAAD. "We observed that treatment of healthy mice with supra-pharmacological doses of an NO donor induced TAAD similar to that seen in Marfan mice. The NO donor treatment also reproduced the degeneration of the aortic wall, an essential step in the development of TAAD," added Redondo. "Through these experiments, we showed that elevated production of NO is necessary and sufficient for the development of TAAD in Marfan Syndrome."
Once the researchers revealed the role of NO in TAAD, they investigated whether they could manipulate the pathway. They focused on two enzymes regulated by NO, soluble guanylate cyclase (sGC) and type I cGMP-dependent protein kinase (PRKG1) which are both found at abnormally high levels in Marfan patients and the mouse disease model.
"We were able to completely reverse the aortic disease in Marfan mice by treating them with inhibitors of these two proteins or by genetically silencing the expression of Prkg1, demonstrating that the NO-sGC-PRKG1 pathway mediates the development of TAAD in Marfan Syndrome," revealed Campanero.
"The results of this study open the way to the use of sGC and PRKG1 inhibitors in preclinical and clinical trials for this syndrome and possibly other aortic diseases," said Redondo.
High NO levels were also linked to molecular changes in the blood that could serve as biomarkers of disease.
"Working with the CNIC Proteomics Unit and clinical groups at Vall D´Hebron, Puerta de Hierro, Marqués de Valdecilla, and Ghent University hospitals, we found that the high NO levels in Marfan Syndrome lead to increases in protein nitration and cGMP in the blood of mice and patients with this disease," said Campanero.
"This discovery has important implications for patients with this syndrome because these molecules could be used as biomarkers for disease monitoring, and we are now studying their potential as prognostic indicators," explained Redondo.
Sources: AAAS/Eurekalert! via CNIC, Nature Communications