After the heart muscle cells of a newborn develop to a certain point where the body has seemed to consider is sturdy enough to support life, the chromosomes of these cells wear away, a process that occurs very quickly after birth. A team of scientists at the Spanish National Center for Cardiovascular Research (CNIC) are picking apart the disappearing act pulled by newborn cardiomyocytes, hypothesizing that the resurrection of these chromosomes later in life could help adult hearts struggling with repair following damaging cardiac events.
After cardiomyocyte chromosomes wear away, these cells are limited in their ability to multiply and replace damaged tissue. For adults at risk for heart failure, stroke, and even death following a heart attack, gaining back cardiomyocytes’ capability early in human life to self-proliferate could be the difference between life and death.
CNIC researchers hypothesized that the initial disappearance of cardiomyocyte chromosomes is connected to a disconnect between telomeres, repetitive and protective sequences of DNA found on each end of chromosomes like the cap on a water bottle, and the enzyme that helps to produce them.
Without this enzyme, called telomerase, other cells in the body recognize stunted telomeres as damaged DNA, stopping the cell cycle from continuing to produce more cells. CNIC researchers initially though the lack or mutation of the telomerase enzyme causes the cardiomyocyte chromosomes to disappear early in life, and providing adult cells with intact telomerase could then be the solution to restoring self-replicative abilities in cardiomyocytes.
To test their hypothesis, CNIC researchers first examined the length of telomeres in newborn mouse cardiomyocytes, and they found that the mice telomeres quickly disintegrated by the time the mice reached a week of life. They simultaneously saw both a decrease in telomerase expression and activation of the DNA damage response that halts the cell cycle as well as activation of a cell cycle inhibitor molecule called p21.
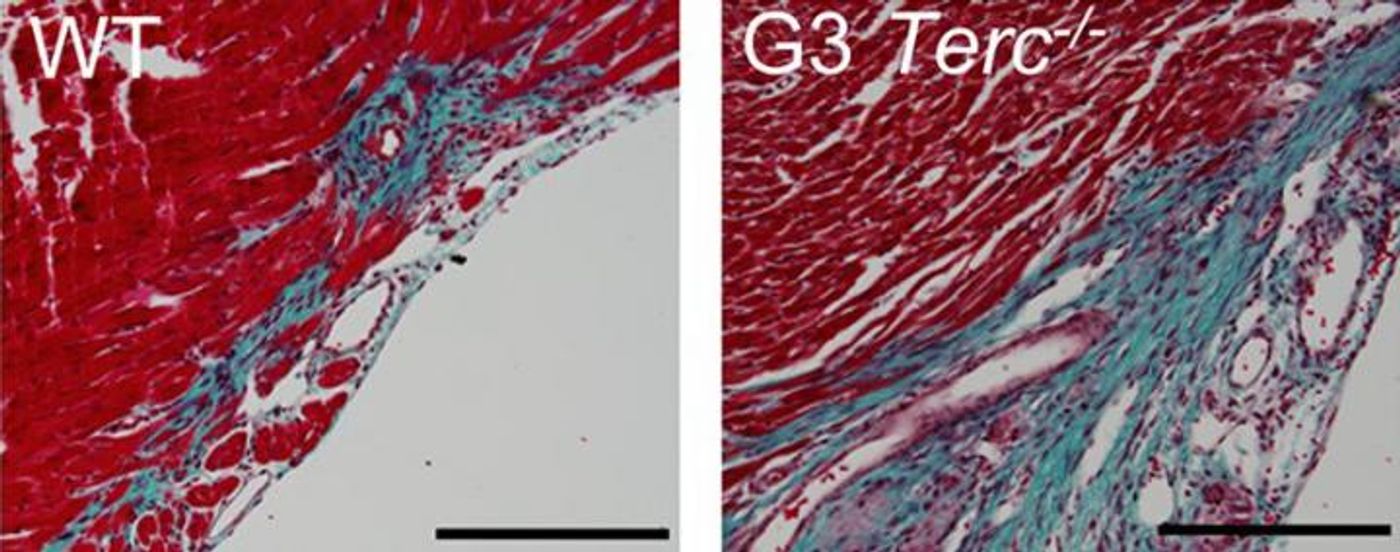
Next, they experimented with telomerase-deficient mice. The cardiomyocytes of these mice stop proliferating just one day after birth. When the mice experienced cardiovascular damage the second day after birth, muscle tissue failed to regenerate, while mice with normal levels of telomerase responded as expected, by self-proliferating new muscle tissue.
Lastly, the researchers tested the regenerative capacity of cardiomyocytes when the cell cycle inhibitor p21 was knocked out. These mutants showed an enhanced ability to regenerate cardiomyocytes, as p21-deficient mice repaired damaged cardiac tissue on their own more effectively than normal wildtype mice of the same age.
The take-away is this: manipulating and/or maintaining cardiomyocyte telomere length could boost the regenerative capacity of adult cells, helping people to heal themselves after a heart attack to prevent the dangerous aftermath that can come from this type of dangerous cardiac event. The CNIC researchers next plan to develop telomerase overexpression mouse models to see if they can extend the “regenerative window.” Will they be successful? Only time will tell.
Their current study was recently published in
The Journal of Cell Biology.
Sources:
Rockefeller University Press,
YourGenome.org