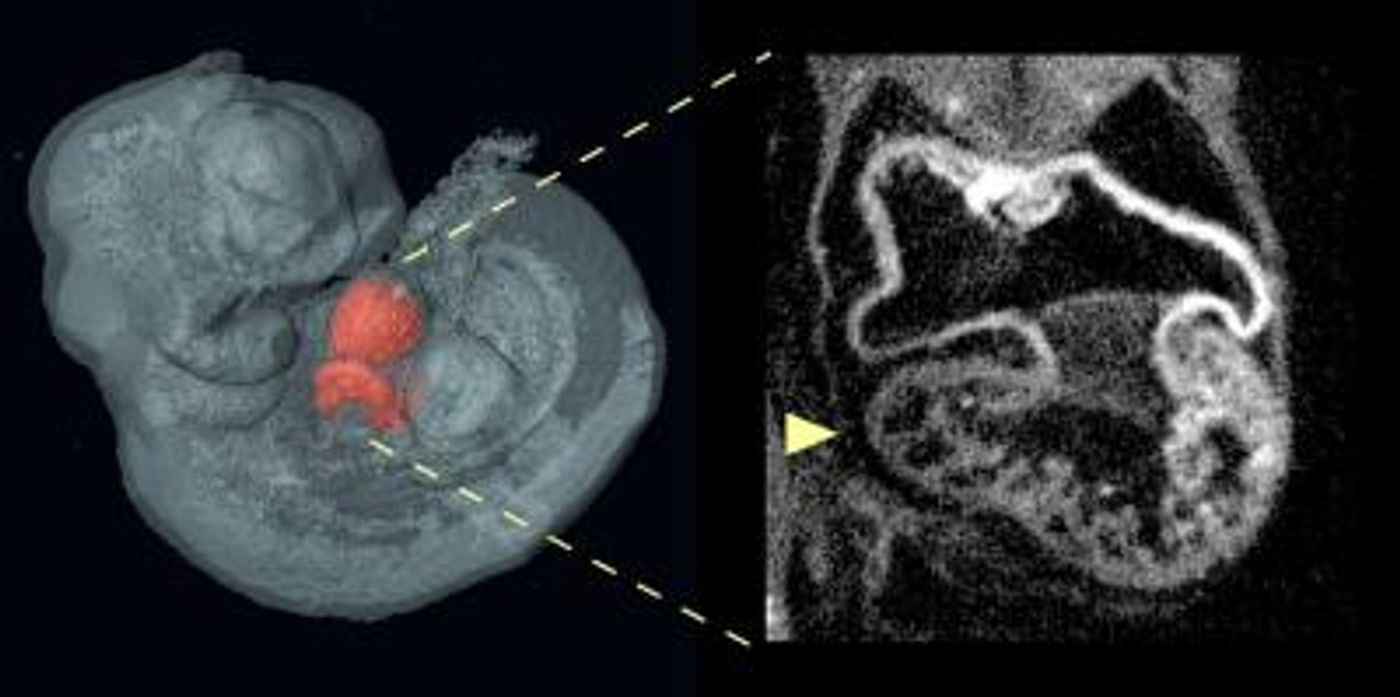
Scientists have reason to believe that most congenital heart defects are a result of a painfully complicated web of genetic connections, rather than a simple one-solution answer. In a novel study from the University of California - Irvine (UCI), researchers demonstrate for the first time how major risk factors for heart defects are likely to stem from outside of the heart itself.
As a leading cause of birth-defect related deaths, congenital heart defects (CHD) are of major concern to doctors and scientists, who are trying to understand how they occur and how they can be prevented or treated as early as possible. “Our results lead us to hypothesize that heart defects such as [atrial septal defects] occur when the heart does not grow quickly enough to meet the demands of the developing body,” explained Anne Calof, UCI professor.
Calof, along with Arthur Lander and colleagues, focused on atrial septal defects (ASDs) in mouse models of Cornelia de Lange Syndrome (CdLS), a developmental disorder that often coexists with heart defects like ASD. Individuals with CdLS experience slow growth during embryonic development and after birth, most often resulting in short stature, intellectual disability, and abnormalities of the bones in the arms, hands, and fingers. In addition to being stricken with heart defects, people with CdLS often have problems with seizures, eye problems, cleft palate, microcephaly, hearing loss, and digestion issues.
The team from UCI began their investigation with a gene called Nipbl. For both mice and humans, CdLS is usually caused by any mutation that inactivates a single copy of Nipbl. Nipbl controls the expression of hundreds of genes from all over the body, so the loss of activity from just one Nipbl copy ignites a chain reaction of damage.
For mice with CdLS and with these types of Nipbl mutations, they have about a 30 percent chance of also having atrial septal defects.
They experimented with mice models of CdLS by either introducing or eliminating Nipbl mutations in different tissues as the embryos were developing. The results weren’t black and white; there wasn’t one tissue or one gene that seemed to be at the center of disease causation. In fact, Nipbl deficiency in some body tissues actually had a protective effect against the development of atrial septal defects.
Rather, heart defect development appeared to be controlled by intricate interactions between gene activity in tissues of the heart and tissues from all over the body. The team from UCI also believes that this is probably the case for other types of congenital heart defects.
“What we learned from Nipbl-deficient mice may actually be more typical of the way most CHDs arise,” said Arthur Lander, director of UCI’s Center for Complex Biological Systems.
This study was recently published in the journal PLOS Biology.
Sources:
University of California - Irvine, U.S. National Library of Medicine
Images: Benedikt Hallgrimsson/University of Calgary, ehjcimaging.oxfordjournals.org