According to the Centers for Disease Control and Prevention, most of the 20 million adults in the United States with chronic kidney disease will die from heart failure before they reach the lethal stage of kidney failure. The reason for this particular case of heart disease? Calcification of the arteries.
Osteoblasts are specially-designed mesenchymal cells that deposit calcium in the bone, building them up and making them stronger. While calcification in the bones is normal and healthy, osteoblast-directed calcification of the arteries leads to inflammation, hypertension, and a buildup of plaque in the blood vessels, a condition called atherosclerosis.
"We expect to find osteoblasts in bone, not blood vessels," said senior author of the recent Cell Stem Cell article Benjamin D. Humphreys, MD, PhD.
However, it’s not directly the fault of osteoblasts that calcification destroys the arteries and leads to heart failure. Osteoblasts are derived from connective tissues that are also a precursor to fat tissue and smooth muscle. In fact, smooth muscle cells actually heal blood vessels, improving arterial flow and contraction. So how does the signal get contorted in such a way that osteoblasts are formed in the wrong place at the wrong time?
A new study from the Washington University School of Medicine identifies the cell type responsible for cardiovascular disease in connection to chronic kidney disease: Gli1 positive stem cells. These are the adult stem cells that have the flexibility of becoming many types of connective tissues: smooth muscle, fat, and osteoblasts.
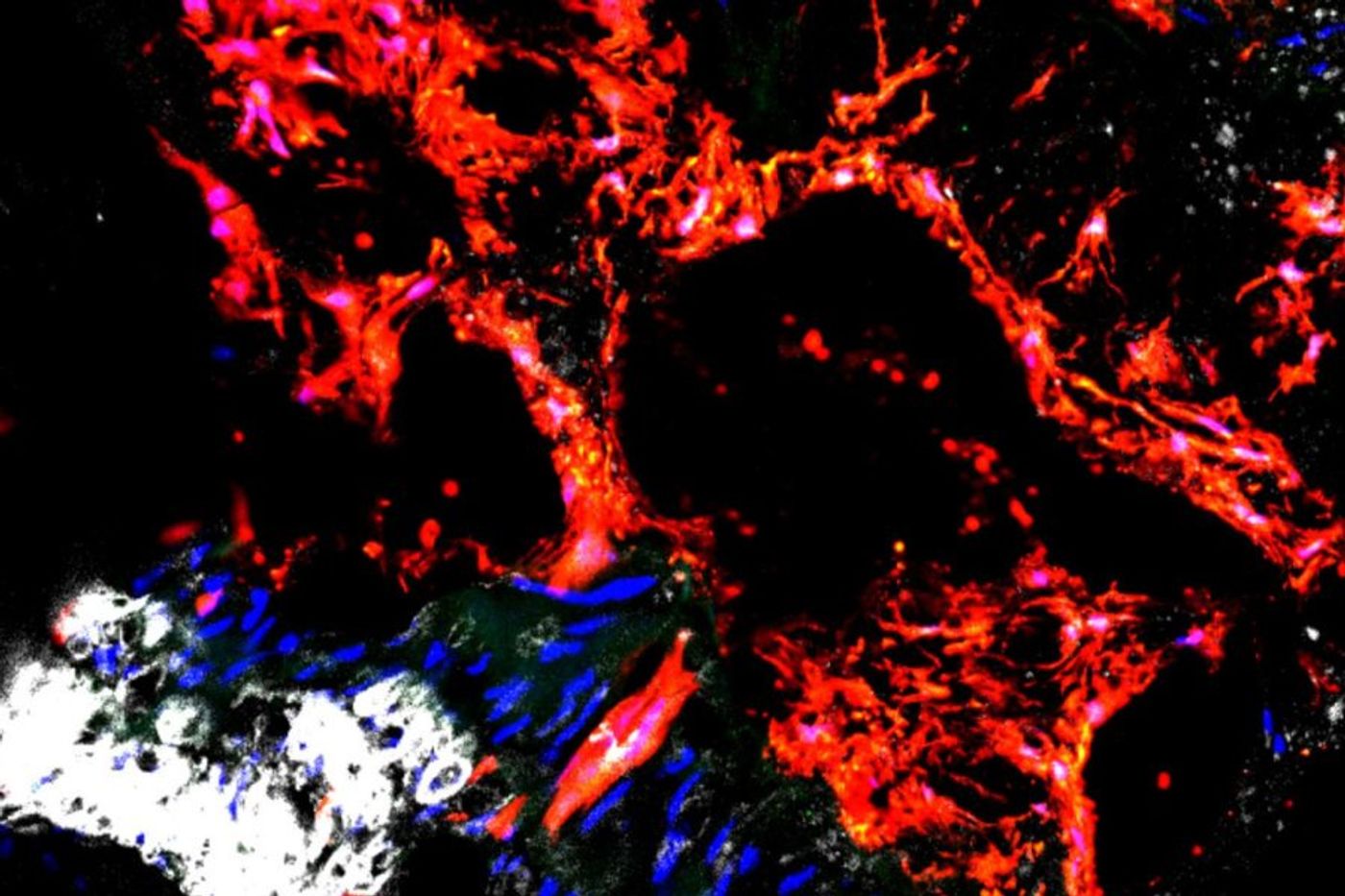
Researchers examined mice with chronic kidney disease, and it appeared that the diseased environment caused the Gli1 stem cells to differentiate into osteoblasts in the arteries rather than smooth muscle. This cellular error only exacerbated the harmful condition the mice were already experiencing due to the kidney disease.
These cells may be trying to perform their healing role in responding to injury signals, but the toxic, inflammatory environment somehow misguides them into the wrong cell type,” Humphreys explained.
Their findings from the mouse studies were confirmed later during analysis of donated tissue from patients who died of kidney failure. "We found Gli1 cells in the the calcified aortas of patients in exactly the same place we see these cells in the mice," Humphreys said. "This is evidence that the mice are an accurate model of the disease in people."
Additionally, they found success in preventing calcification of blood vessels in adult mice when they surgically removed Gli1 cells. Future treatments may build off of Gli1 removal or drugs targeting the stem cells, but scientists will have to be careful not to damage the Gli1-derived smooth muscle cells that benefit arterial flow.
"Now that we have identified Gli1 cells as responsible for depositing calcium in the arteries, we can begin testing ways to block this process," Humphreys said. "A drug that works against these cells could be a new therapeutic way to treat vascular calcification, a major killer of patients with kidney disease.”
Sources:
Washington University School of Medicine,
Acta Reumatólogica Portuguesa