Hypoxia: A fundamental part of cardiac development
The hypoxia response has been found to be vital for the proper formation of the heart ventricles during gestation, adding an additional stage to embryonic development. A new study from the Centro Nacional de Investigaciones Cardiovasculares Carlos III (CNIC) investigates the fundamental hypoxia response outside of its role in cancer and tumor metastasis.
Hypoxia, or a sudden reduction in oxygen levels, triggers a complex adaptive response to reestablish the supply of nutrients and oxygen to the tissues.
Before this study, as far as researchers knew, heart metabolism is controlled by glucose consumption during embryonic development and fatty acid oxidation after birth. Soon after birth, the control from glucose transitions into the “energetically more efficient” fatty acid oxidation, where more oxygen is available after birth than during gestation. However, as scientists from the current study discovered, there is a new point of regulation during gestation: a decrease in the cardiac levels of hypoxia inducible transcription factor 1 (HIF-1).
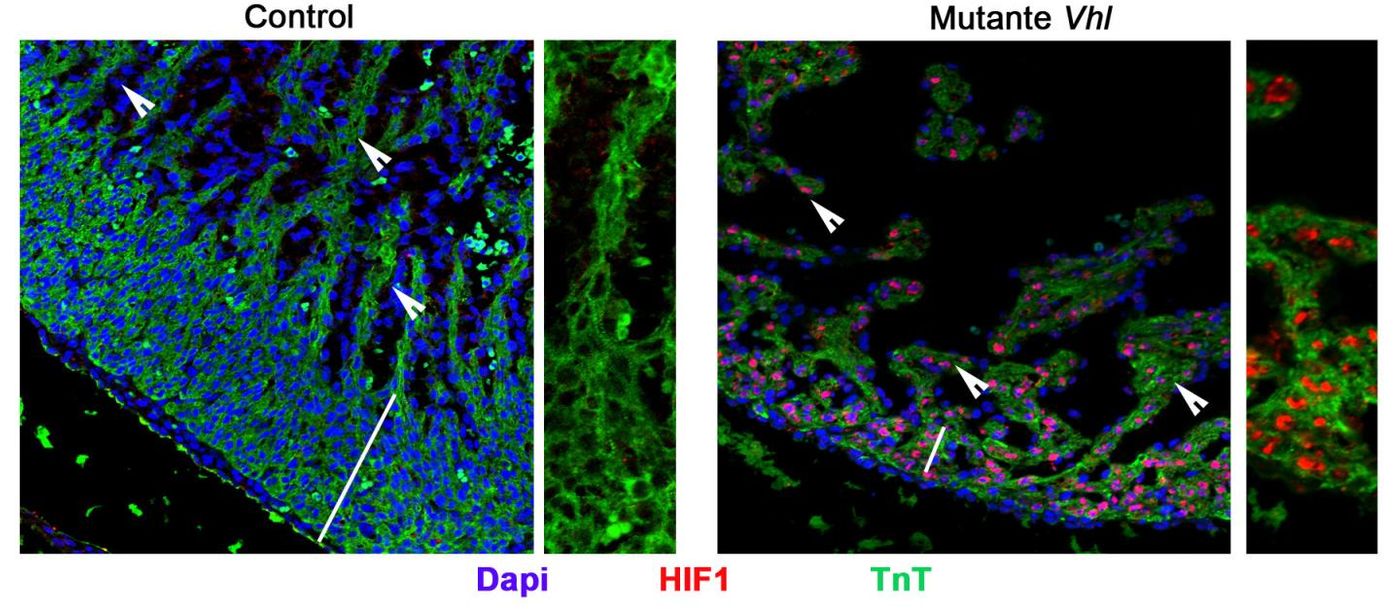
They found that high levels of HIF1 and glycolytic metabolism defined the compact myocardium, where contractile myocytes developed. On the other hand, low levels of HIF1 and glycolytic enzymes are found in the trabecular myocardium, which houses the ventricular conduction system for transmitting cardio-electric stimuli.
Understanding the dispersion of HIF1 during cardiac development could help researchers identify mechanisms underlying some congenital heart conditions like preeclampsia and gestational diabetes and also for diseases of the adult heart, like myocardial infarction. This condition is caused by a lack of oxygen, which induces HIF1 activation. This could “reprogram the metabolism of the mature myocardium toward an embryonic pattern,” explained study leader Dr. Silvia Martin-Puig.
Martin-Puig and his team studied heart development in mice at midgestation. The metabolic separation between the compact myocardium and the trabecular myocardium was seen to occur with a sharp drop in the expression of glycolytic enzymes, an increase in the number of mitochondria and the activity of genes involved in fatty acid metabolism, and a loss of HIF1 expression in the heart.
Additionally, maintaining HIF1 activation beyond midgestation causes severe structural defects incompatible with life. “The sustained presence of HIF1 maintains a metabolic program based on glucose consumption and causes a sharp decline in the number and activity of cardiac mitochondria, impeding the switch to oxidative metabolism,” said Martin-Puig. “These alterations to the metabolic program and HIF1 expression in the embryo compromise the contractile capacity of the myocardium and significantly impair heart function."
Understanding the role between dwindling HIF1 levels and healthy maturation of the heart muscles could help scientists in the future treat diseases where the heart’s chambers do not develop correctly.
This study was recently published in the journal Development Cell.
Source: Centro Nacional de Investigaciones Cardiovasculares Carlos III