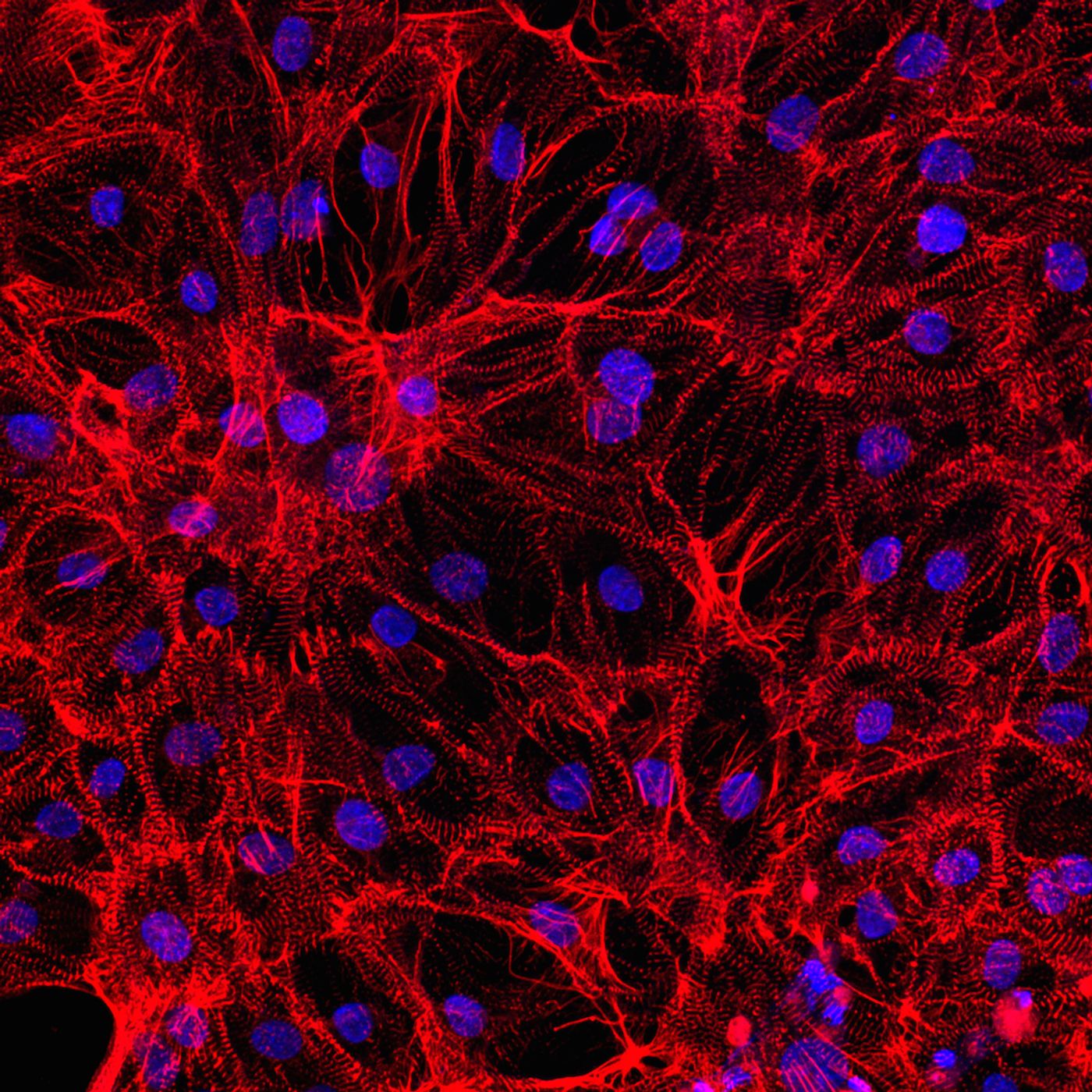
Success In Growing Mature Heart Cells from Stem Cells
Whether to make actual repairs on the heart or just to study the diseases that plague it, many scientists are in hot in pursuit of the ability to transform stem cells into mature, functional heart muscles cells. The most recent successful venture in this arena comes from researchers at Johns Hopkins University, who grew stem cells to maturity in newborn rat hearts.
"The hope is that our work advances precision medicine by giving us the ability to make adult cardiomyocytes from any patient's own stem cells,” explained Chulan Kwon, PhD, who led the study. “Having that capability means having a way to test each patient for old and new drug sensitivities and value and to have a scalable process to create large cell sources for heart regeneration."
Among their stem cells growing in the lab, Kwon and his team observed that they were not expressing the genes necessary for their transition into a mature, adult state. After looking closer at the hearts of newborn rats, they saw that the same genes lacking expression in the lab stem cells were activated in the newborn rat hearts. What this means is the genes failing to turn on this particular set of genes wasn’t a matter of age, it was a matter of something missing in the lab culture.
In an attempt to fix this deviation, the team developed a cell line of immature heart cells taken from mouse embryonic stem cells, tagged them with a fluorescent protein so they could tell them apart from host cells, and injected 200,000 of the cells into the ventricle of newborn nude rats.
Nude rats have deficient immune systems, and were used in this experiment so the rats would not reject the mouse embryonic stem cells. After one week, the mouse embryonic stem cells were still immature, but after one month, the mouse embryonic stem cells looked like adult heart muscle cells.
After the mice embryonic stem cells had appeared to have transformed into functional mature cells, researchers compared 312 genes in the individual mouse cells grown in the rats hearts to similar genes found elsewhere:
- Genes found in immature human heart cells
- Genes found in adult human heart cells
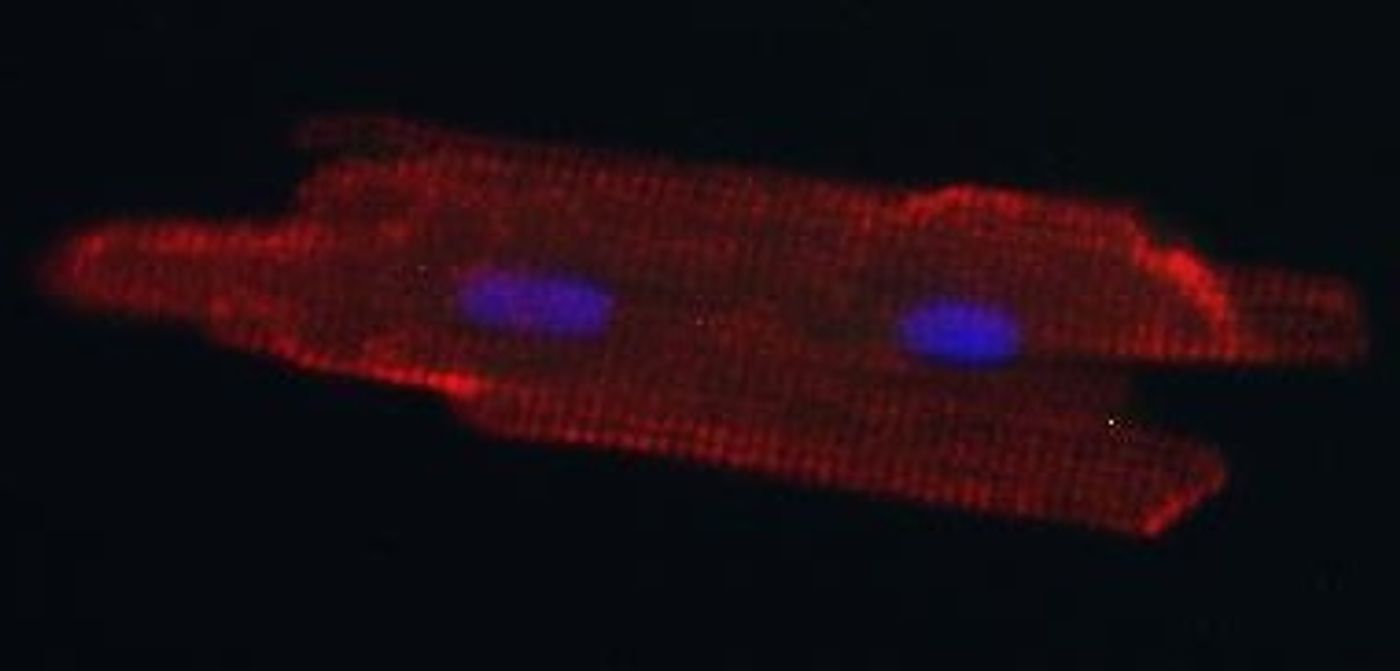
They quickly saw that the mouse embryonic stem cells grown into mature muscle cells in the rat hearts had more in common with genetics of adult heart muscle cells than with immature heart cells. Then, an optical microscopy experiment confirmed that the mature muscle cells grown in rat hearts could contract like normal heart muscle cells.
Image: An adult heart muscle cell was grown in a newborn rat heart. Credit: Kwon lab
Their next study was of human adult skin cells obtained from a healthy human donor which were transformed into induced pluripotent stem cells in the lab through a chemical conversion process. After injecting these stem cells into newborn rats heart, they matured within one month.
Lastly, the researchers again obtained induced pluripotent stem cells, this time from a patient with a heart condition called with arrhythmogenic right ventricular cardiomyopathy (ARVC). This condition is passed down from parent to child and is a leading cause of sudden death in young adults. However, the genetic mutation responsible for the disease is only evident after heart cells mature.
After one month, induced pluripotent stem cells from a human ARVC patient that were injected into the ventricle of a rat heart began to resemble heart tissue of a patient with ARVC. Specifically, the researchers saw a massive collection of fat, and the cells were dying faster than healthy cells could replenish them.
Kwon’s method of growing stem cells in newborn rat hearts was shown to be very successful in his series of studies. It seems that these heart hosts contain the necessary biological and chemical signals for the implanted stem cells to grow and overcome the “developmental blockade” that normally stops their growth in the lab.
In addition to boosting advanced studies of heart disease pathology, designing new diagnostic tools, and developing new treatments, Kwon is hopeful that his studies will be expanded to other areas of stem cells research, which he said “really opens up a new avenue to getting stem cells to mature.”
Kwon’s study was recently published in the journal Cell Reports.